A drug to treat metastatic, triple-negative breast cancer is being tested in a national clinical trial at the UAB Comprehensive Cancer Center, where it was developed.
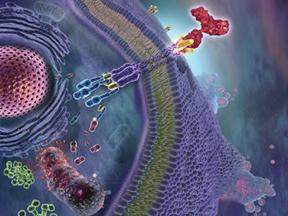 |
| Image Credit: Echo Medical Media. Not for reuse. |
Tigatuzumab is a monoclonal antibody developed by Tong Zhou, M.D., associate professor of medicine and cell biology and scientist at the Comprehensive Cancer Center, in partnership with Daiichi-Sankyo Co. Ltd. of Japan. This laboratory-produced molecule binds to tumor-cell receptors and sends the cell a death signal.
Tigatuzumab appears to be most effective against triple-negative breast cancer. These aggressive, difficult-to-treat tumors comprise about 25 percent of all breast cancers and most often occur in young African-American and Hispanic women.
UAB is the lead institution for the nationwide clinical trial, which launched March 3, 2011, and includes 15 members of the Translational Breast Cancer Research Consortium. UAB Comprehensive Cancer Center senior scientist Andres Forero, M.D., is principal investigator. The trial is open to any patient age 19 and older with metastatic, triple-negative breast cancer.
“All the pre-clinical evaluations suggest that the new antibody will be effective against triple-negative breast-cancer cells and that may translate into a new treatment,” Forero explains.
Much of the early work involving Tigatuzumab was supported by a $6.4 million Susan G. Komen for the Cure Promise Grant, co-funded by the Triple Negative Breast Cancer Foundation, which was awarded in 2009 to Forero for this research. The Birmingham-based Breast Cancer Research Foundation of Alabama also provided seed money for the drug’s development.
“Support from these foundations accelerates progress in the fight against triple-negative breast cancer. This trial is another example of the UAB Comprehensive Cancer Center’s ability to quickly move leading-edge discoveries from the laboratory to patients,” Forero says.