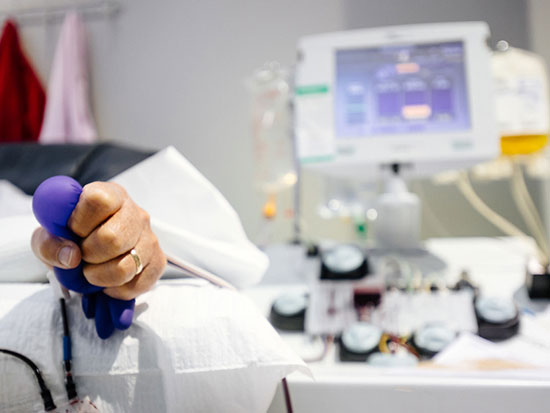
 Donations of plasma from patients recovered from COVID-19 are being used to boost the immune systems of newly infected individuals using convalescent plasma therapy, one of three treatments being studied in new outpatient clinical trials at UAB.It is the call no one wants: On the other end of the line is a doctor telling you that you have been diagnosed with COVID-19 or a loved one who has tested positive.
Donations of plasma from patients recovered from COVID-19 are being used to boost the immune systems of newly infected individuals using convalescent plasma therapy, one of three treatments being studied in new outpatient clinical trials at UAB.It is the call no one wants: On the other end of the line is a doctor telling you that you have been diagnosed with COVID-19 or a loved one who has tested positive.
What can you do now? (If you are a UAB employee or student, you should report a potential high-risk exposure, and a positive test, if it was not conducted at UAB, on your daily Healthcheck. But what then?)
For the first several months of the pandemic, there has not been much that doctors could do for patients outside the hospital, said Turner Overton, M.D., director of UAB's outpatient COVID-19 clinic and professor in the Division of Infectious Diseases. Inpatients at UAB can receive the antiviral drug remdesivir or the steroid dexamethasone if they are having difficulty maintaining sufficient oxygen levels. UAB is participating in several other studies of treatments that are open to inpatients. But there have been no targeted treatments for patients diagnosed with COVID-19 if they are not sick enough to be hospitalized.
"We have all these people who are having symptoms, and we really have very little to offer them, unfortunately," Overton said. "It's really symptom management to help people get through and try to prevent hospitalization or identify those people who will need hospitalization so they can get access to remdesivir or dexamethasone as early as possible."
But now three new clinical trials are underway at UAB that make experimental therapies available to people in the first days after symptoms begin. One is even testing a preventive treatment for people who have been exposed to the disease but do not have a COVID-19 diagnosis.
Transferring immunity
"When we think about ways to treat COVID-19 we can either target the virus using antivirals, such as remdesivir, or we can use the immune system to target the virus," said Sonya Heath, M.D., professor in the Division of Infectious Diseases. "The studies we are enrolling for now use the immune system to block the virus and help to eliminate it.”
How to enrollTo learn more about participating in any of the trials discussed here, or to find out if you are eligible, call (205) 934-6777. |
It is now clear that “the patients who do better with COVID-19 are the ones who generate an immune response to the virus,” Heath said. “They have a shorter course of illness and their viral load is lower. [Viral load is a measure of the amount of virus particles in an infected person’s body.] We know that generating an immune response to the virus is a good thing and decreases your symptoms and your severity. Each of these studies is looking at ways to transfer an immune response to you in some form."
Who can participate?
Who is eligible to participate in these trials? It all depends on where a person is in the course of their infection — or if they have a high-risk exposure to someone with COVID-19.
 If it has been eight days or less since symptoms started:
If it has been eight days or less since symptoms started:
Convalescent plasma outpatient study: With convalescent plasma, "we are taking plasma — the liquid portion of the blood — from somebody who has recovered from the virus and transferring that immunity to someone actively infected with the virus now," said Heath, who is principal investigator for this study. "We've been doing this since early summer for inpatient care and now we are using it with outpatients." The inpatient study showed that plasma with high concentrations of antibodies against COVID-19 was most beneficial, Heath said.
The new outpatient study will treat half of the patients enrolled with an intravenous (IV) infusion of plasma that has high antibody concentrations. The other half of study participants will receive a placebo — plasma from someone who has not had COVID. "We are asking the question: If you give high concentrations of antibody is that beneficial in decreasing severity of illness and decreasing the likelihood that somebody will be hospitalized?" Heath said.
Learn more about convalescent plasma therapy and the trial in this story from the UAB Reporter.
If it has been 10 days or less since symptoms started, or seven days or less since you had a positive COVID-19 test:
 Instead of using antibodies from donors who have recovered from COVID-19, several pharmaceutical companies have identified very strong antibodies against the virus and are making them in their labs. The companies Regeneron and Eli Lilly have each developed their own synthetic antibodies, known as monoclonal antibodies. "The preliminary work from both of these shows that they are effective," Overton said. "If we can get people these monoclonal antibodies early on in their illness, we can really mitigate the long-term effects of COVID-19. If we can stop the virus in its tracks in the first three or four days then we can potentially block the consequences."
Instead of using antibodies from donors who have recovered from COVID-19, several pharmaceutical companies have identified very strong antibodies against the virus and are making them in their labs. The companies Regeneron and Eli Lilly have each developed their own synthetic antibodies, known as monoclonal antibodies. "The preliminary work from both of these shows that they are effective," Overton said. "If we can get people these monoclonal antibodies early on in their illness, we can really mitigate the long-term effects of COVID-19. If we can stop the virus in its tracks in the first three or four days then we can potentially block the consequences."
Regeneron trial
Regeneron's experimental therapy was given to President Trump during his recent hospitalization. "This is a cocktail of antibodies — a combination of two different antibodies targeting proteins on the surface of the virus," Heath said. "If it can bind to the virus it can help eliminate it and reduce the severity of the illness."
Do you have to live in the Birmingham area to take part?No, Heath said. "Many of these studies have been designed to limit the number of in-person visits," she explained. "For example, with the convalescent plasma study we see people on day 1, day 14, day 28 and day 90 and everything else is done by phone. For the Regeneron trial you get one infusion and then you have a few in-person visits but a lot of it is done by phone or a log that you keep." |
Participants in the Regeneron Phase I outpatient trial at UAB are randomly chosen to receive a high dose of the antibody cocktail, a low dose of the antibody cocktail or a placebo. "It is a one-time infusion," Heath said. "There are significant reductions in the viral load with both the low-dose and the high-dose infusions."
Although the UAB trial site is just opening, Regeneron has released early reports on the first 275 patients who enrolled in the trial at other locations, and these included "some beneficial outcomes," Heath said. These include reduce viral levels and improved symptoms, according to the company.
ACTIV-2 (Lilly) trial
This trial is testing a monoclonal antibody developed by Eli Lilly. "Participants will receive either placebo or the monoclonal antibody from Lilly that attaches to the virus and blocks it from infecting cells and causing disease," said Overton, who is principal investigator at UAB for the trial. "Compared to the Regeneron trial this monoclonal antibody has not gotten as much press but it is a similar monoclonal antibody that has been shown to be effective in the preliminary Phase I and II trials."
"We have a great deal of experience using this immune-based approach," Overton said. "Furthermore, it's a proof of concept for vaccines. If the passive immunization strategy works with either convalescent plasma or with monoclonal antibodies, it really gives a lot of strength to pursuing effective vaccines."
 Infectious diseases specialists Sonya Heath, M.D., and Turner Overton, M.D., explain the new trials to the media in this screenshot from UAB's virtual press conference.
Infectious diseases specialists Sonya Heath, M.D., and Turner Overton, M.D., explain the new trials to the media in this screenshot from UAB's virtual press conference.Story continues after box
 If it has been four days (96 hours) or less since you were in close contact with someone diagnosed with COVID-19:
If it has been four days (96 hours) or less since you were in close contact with someone diagnosed with COVID-19:
People who have been closer than six feet from a person diagnosed with COVID-19 for more than 15 minutes during a 24-hour period are considered to be "close contacts." If you meet these criteria, you may be eligible to take part in a UAB study testing convalescent plasma therapy as prevention.
The idea is that an infusion of antibodies from a recovered COVID-19 patient could give your body a head start in fighting off the disease and reduce the risk of having serious complications from COVID-19.
"We have some people who live in a household where they don't have multiple bathrooms and it is hard to quarantine away from elderly parents," Heath said. "They might try to get one person in the household treated with the outpatient treatment [either convalescent plasma or the Regeneron or Lilly monoclonal antibody trials] and then other people in the household who are COVID negative could take part in the prevention phase to really try to see if we can intervene on the severity of illness in their family."
Story continues after box
‘Consider participating in these trials’
After a COVID-19 diagnosis, "people are scared and they don't know what to do — sometimes not doing anything when you feel sort of bad but not terrible seems like a reasonable approach," Overton said. “But we don’t know who is going to get worse disease. We know there are certain individuals — older people, people with certain comorbidities — who are at greater risk for severe disease, but I can tell you that we see healthy people getting severe disease as well. So we are really encouraging anybody who is interested to get involved. There may be some benefit for them and without treatments and vaccines we will be stuck in this situation that we are in."
