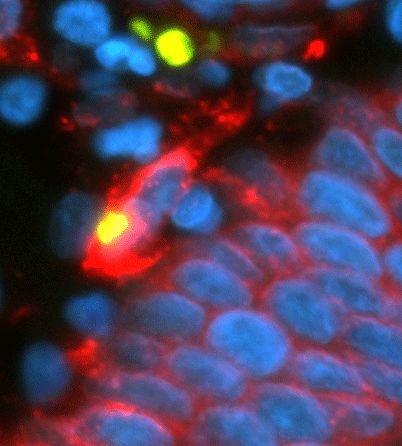
 What's that ... a meteor blazing past a molten planet? No, it's a self-destructing cell just shed from a gastric gland made up of the tightly packed blue cells running across the bottom.
What's that ... a meteor blazing past a molten planet? No, it's a self-destructing cell just shed from a gastric gland made up of the tightly packed blue cells running across the bottom.Gastric glands that secrete digestive juices into the stomach, like all epithelial cells (skin, lining of blood vessels, etc.), constantly shed old cells from their outer layers and replace them with new ones. The turnover keeps tissues viable throughout adult life. When the shed cells sense they have outlived their usefulness, they initiate self-destruct mechanisms.
With some cells always in the process of self-destructing, other nearby cells are charged with swallowing up the dying cells and disposing of them. That explains the red cloud surrounding the yellow dot at the center of the image. An antigen-presenting cell (dyed red) has engulfed a self-destructing cell (yellow) to remove it.
The system works pretty well until a person's stomach gets infected with the bacteria Helicobacter pylori.
The infection causes cells to release TNF-alpha, a signaling chemical that triggers the waves of cell activation and chemical release meant to kill invading bacteria or viruses (inflammation). While the process protects us against infectious disease, it also plays a role in many disease processes when it goes too far.
A recent study by a team of UAB researchers found that, along with triggering inflammation, TNF-alpha also blocks the engulfment and removal of dying cells. Cellular debris builds up to drive further inflammation in a vicious cycle.
Why does this matter? Inflammation caused by Helicobacter pylori is behind the development of nearly all ulcers. Furthermore, the same chronic inflammation damages DNA, creating risk for hard-to-treat gastric cancer.
The image was created by Diane Bimczok, DVM, Ph.D., an instructor in UAB's Division of Gastroenterology and Hepatology, using routine fluorescence microscopy and digital imaging. Phillip Smith, M.D., was senior author of the related study.
This post originally appeared on The Mix, UAB's science blog.