In a discovery many neurobiologists once thought too farfetched to accept, a protein known to help sperm move toward eggs during conception of the worm C. elegans appears to play a central role in Lou Gehrig’s disease, according to a University of Alabama at Birmingham study published today in Developmental Cell.
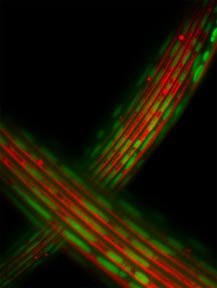 |
| In healthy muscle cells, energy-producing mitochondria (in green) line up along elements (in red) that need energy to flex. Lou Gehrig's disease may disrupt these neat rows. |
The results cap a seven-year effort by a team of researchers to test whether an ancient, unusual cell-signaling mechanism might explain a mysterious, often fatal disease. Specifically, the work explores the idea that major sperm protein has cell-signaling roles in many cell types across many species — including human nerve cells — that use it to help their muscle-cell partners thrive.
“We began as basic scientists looking at how worm cells signal each other, but came across something else we thought was important and couldn’t let it go,” said Michael Miller, Ph.D., associate professor of cell biology at UAB and corresponding author for the study. “Our results provide new detail explaining how defective MSP signaling could contribute to nerve- and muscle-cell failure in Lou Gehrig’s disease and, we hope, lay the foundation for future drug design.”
Also known as amyotrophic lateral sclerosis, Lou Gehrig’s disease causes muscle cells that control movement to fail, including those that enable breathing. Patients become paralyzed, and most die of respiratory compromise within three years. There are no treatments, and until now, no plausible explanation of how the disease specifically affects motor neurons and the muscle cells they innervate.
Another mystery surrounding a disease that has confounded medical science for 150 years is that it almost always strikes adults ages 40-60. The study results suggest that the MSP-signaling pathway operates continuously as we age and disruptions make adults susceptible to ALS. The reason 90 percent of ALS cases arise in people with no family history also remains elusive. The many questions highlight the importance of any clue, like the fact that all known cases of ALS, inherited or not, come with reduced MSP levels, researchers said.
A long, strange trip
The current study has its roots in a 2001 paper in which Miller and colleagues shocked “the worm (research) world,” concluding that, along with its known role of enabling sperm cells to “crawl” toward eggs during reproduction, major sperm protein also is secreted by sperm cells like a hormone. It signals nearby eggs that they should prepare for fertilization. Most known signaling proteins looked nothing like major sperm protein, and many observers thought the results were too unusual to be correct.
In the years since, advances in DNA sequencing changed the accepted perspective on MSP’s origin and prevalence. Though researchers first recognized it as a small protein found only in worm sperm, subsequent work revealed that nature had created it in other forms and built it into larger proteins — and not only in worms. Proteins with the MSP “domain” have been found in many animal cell types, including human nerve cells. No matter where it occurs or what it does, MSP retains its original worm name.
Stranger still, a study in 2004 revealed that a small random change, or mutation, in the MSP part of a human protein called VAPB was responsible for an inherited form of ALS in a large Brazilian family. Miller and his team saw the study and began hypothesizing that the VAPB major sperm protein might be secreted by human nerve cells to promote both their own survival and muscle function.
Meanwhile, Hugo J. Bellen, Ph.D., D.V.M., professor of molecular and human genetics at Baylor College of Medicine and renowned fly neurobiologist, had seen the same paper and decided to investigate the role of VAPB in fruit fly neurons. After meeting during a UAB seminar in 2004, Miller and Bellen collaborated, and in 2008, published a paper in Cell that confirmed the VAPB major sperm protein was secreted by fly and worm cells. They also reported that the ALS-causing VAPB mutation prevented MSP secretion in the study animals. And finally, the study identified MSP in human blood serum, which supported the idea that human cells secrete it.
Tangled mess
For the current study, the Baylor/UAB research team used molecular techniques to halt MSP signaling in adult worms. The team attached glowing tags to the worms’ mitochondria, the “machines” within cells that convert sugars into energy for life processes. They then watched as worms without MSP developed “massive” mitochondrial abnormalities. Mitochondrial defects previously had been observed in ALS patients, but the cause was unknown.
In normal muscle cells, mitochondria localize to the areas most in need of energy, like the contractile fibers that do the work of flexing, and junctions at which nerves and muscle meet. In the mutants with no MSP signaling, mitochondria no longer line up near energy-requiring sites and produce 30 to 40 percent less energy than normal.
The team then showed that secreted major sperm protein affects muscle cells by signaling through two well-known classes of receptor proteins expressed throughout the human nervous system, leukocyte antigen-related (LAR)-like phosphatase and roundabout (Robo) receptors. Genetic changes in these receptors correlate with ALS susceptibility, but how they do so is unknown.
"We propose that motor neurons secrete the VAPB major sperm protein to correctly position mitochondria and to encourage their energy production in muscle cells and possibly in neurons themselves,” said Sungmin Han, first author of the paper and the graduate student responsible for much of the project work in Miller’s lab. “Further, we believe our results help establish mitochondrial dysfunction as a linchpin of ALS, but acknowledge a need for further study, particularly in human cells.”
A former post-doctoral fellow in the Bellen lab at Baylor, Hiroshi Tsuda, M.D., Ph.D., (now at McGill University), as well as post-doctoral fellows Pauline Cottee, Ph.D., and Se-Jin Lee, Ph.D., graduate students Youfeng Yang (now a UAB postdoctoral fellow) and Jessica Winek, and Research Technician Jack Vibbert in the Miller lab within the UAB Department of Cell Biology made important contributions to the work. Claire Haueter from the Bellen lab played a critical role as well.
The work was funded by the Muscular Dystrophy Association, ALS Association, American Cancer Society and Howard Hughes Medical Institute.