
Taking organic chemistry as an undergraduate almost finished my career in biology before it ever started. So, it’s ironic that I am now part of a project that involves both biology and chemistry. Fortunately, I have discovered that the “hands-on” chemistry in this project is remarkably like doing your laundry!
Both Maggie and Sabrina already talked about the part of our project that involves looking for compounds produced by algae, sponges, and tunicates (see “Bags of Water and Cactus Sponge” and “All Inclusive Amphipod Hotels”). These compounds prevent the organisms from being eaten by most (but not all!) small gastropods and crustaceans, and allow them to successfully grow and reproduce in the ocean.
But how do we look for defensive compounds in plants and animals? Well, that’s where Andrew – and the chemistry – come in. Andrew Shilling (pictured below on the left) is a Ph.D. candidate in the laboratory of Dr. Bill Baker (a co-principal investigator on this project; pictured on the right) at the University of South Florida. Andrew has a unique background in both biology and chemistry, which allows him to look at our project from multiple angles. In his own words, “Chemistry is cool because it’s biology on a much smaller scale…and this allows you to see the intricacies of the system you’re studying.”

Let me introduce you the steps it takes to look for defensive compounds. Remember the last time that you dumped coffee all over your nice, white shirt? The entire process of extracting chemicals from plants and animals could be compared to getting that dark stain out of your laundry.

First, Andrew adds the alga (in the picture below he has the alga Plocamium, but it could be any plant or animal) to a small jar containing a solvent, which extracts the defensive compounds from the alga. It’s like adding detergent to your clothes in the laundry, which dissolves and takes the stains out of your clothes. Then, Andrew and Michelle mash up the alga in the solvent to expose more of the algal tissue to the solvent, just like you would rub detergent or stain remover into the stain on your shirt.

After letting the alga/solvent mixture sit for 24 hours, they take the solvent (which now has all the chemicals from the alga), throw away the alga, and run the solvent through a large, white machine called a gas chromatograph (the top of the machine is visible in the picture below). The gas chromatograph produces a picture showing the compounds from the algae that were suspended in the solvent.
On Andrew’s computer screen below, you can see the pictures from two pieces of alga (and here’s where the laundry analogy ends). Each blue peak represents a defensive compound in the solvent. Both pieces of alga (one’s on the top of the computer screen and the other is on the bottom) have three relatively tall peaks (or compounds). But, even though the two pieces are from the same species of algae, the pattern of the peaks (types of compounds) are different. It’s amazing! One algal species can produce multiple sets of compounds, or what we call multiple chemotypes. So far, Andrew and our group have discovered 12 chemotypes in just one algal species!
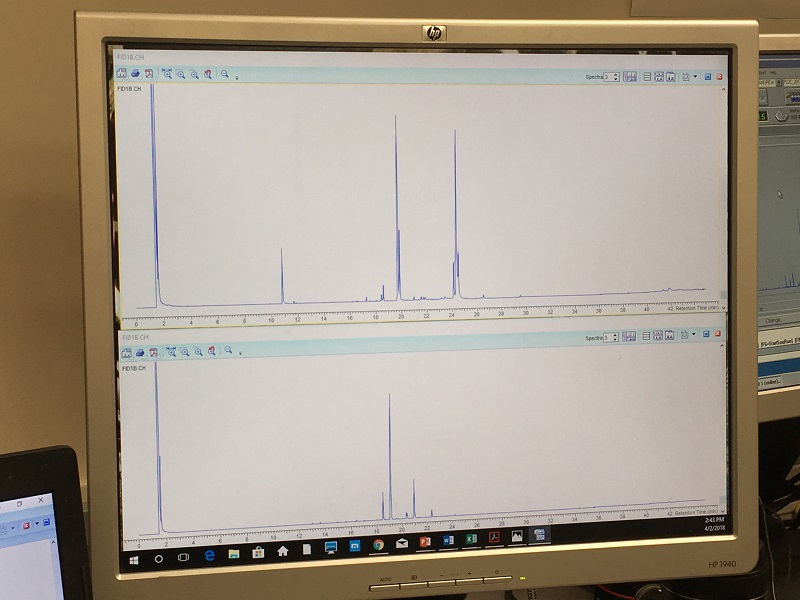
But, why would one algal species produce multiple combinations of compounds? We don’t know! Our group has many ideas. Multiple chemogroups could allow algae to grow in new areas of the ocean, where light, depth, and predators vary. That’s the question we are trying to answer this season. But in the meantime, this chemistry is loads and loads of fun!