 |
Gorgas Case 2026-9 |
 |
|
We hope you enjoyed the 2026 series of live cases from Peru each week. The Gorgas Diploma Course takes place annually in February and March, and we will be in touch at the start of next year's case series. The following patient was seen by Gorgas participants at Hospital Regional in Iquitos.
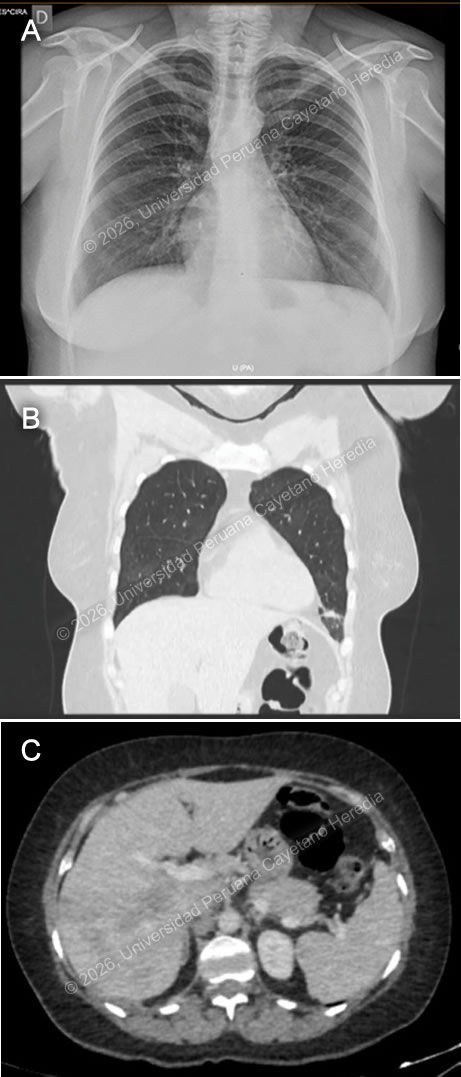
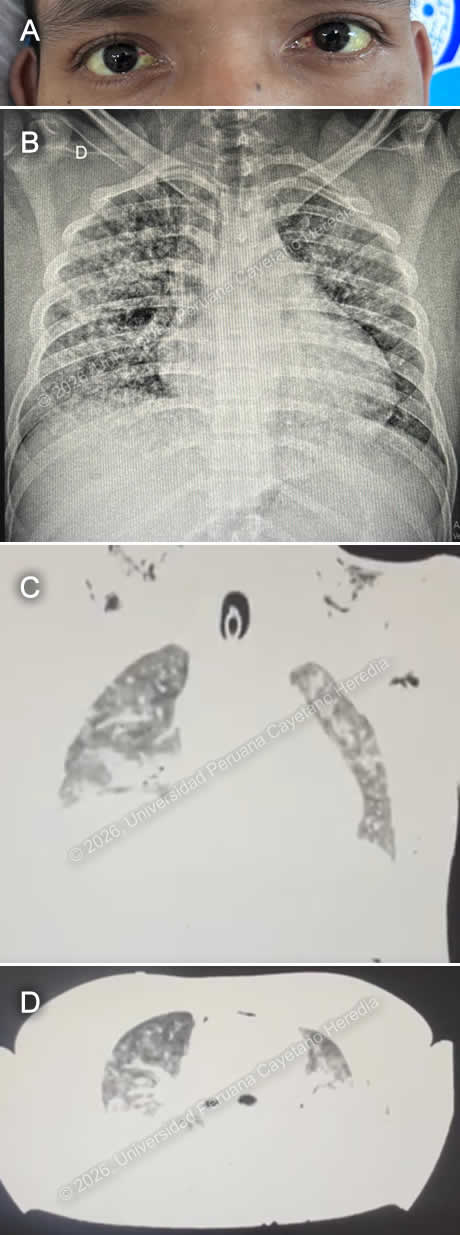 History: This is a 19-year-old male from Iquitos who presented with a one-week history of fever, shortness of breath, and vomiting. One week prior to admission, he developed fever, headache, malaise, chills, and multiple episodes of vomiting, which persisted over the following days. Four days prior to admission, he began experiencing diarrhea and abdominal pain. One day before admission, he developed dyspnea, abdominal distension, and generalized myalgias. On the day of admission, he had multiple episodes of vomiting, initially containing food with streaks of blood, progressing to fully bloody emesis. He also noted reddish discoloration of his urine. He subsequently presented to the emergency department and was admitted. On review of systems, he reports anorexia, shortness of breath, poor oral intake, and conjunctival suffusion. He denies jaundice, skin lesions, retro-orbital pain, and lower extremity edema. Epidemiology: The patient has a history of pancreatitis in 2015 and a prior dengue infection in 2019. He was born in Nauta and currently lives in Punchana, a district of Iquitos. He is a student who also works part-time painting bathrooms and driving a moto-taxi. He reported drinking water under unsanitary conditions while working. He describes exposure to rodents at home, poor sanitation from a neighbor who raises chickens, and frequent mosquito bites. He denies recent travel or contact with sick individuals, reports occasional alcohol use, and denies tobacco or recreational drug use. Physical examination: On admission, his vital signs included a blood pressure of 80/50 mmHg, a heart rate of 146 beats per minute, a respiratory rate of 24 breaths per minute, a temperature of 36.8°C, and an oxygen saturation of 91% on room air. The patient was disoriented, exhibited pallor on skin examination, and had mild scleral icterus with bilateral conjunctival suffusion (Image A). On respiratory examination, he had diffused bilateral crackles. The patient appeared to be in severe pain. His respiratory function deteriorated, leading to placement on mechanical ventilation and admission to the ICU. Laboratory: On admission, laboratory evaluation showed a hemoglobin level of 12.4 g/dL, severe thrombocytopenia with a platelet count of 23,000/µL, and leukocytosis of 13,000/µL with 86% neutrophils, 4.54% lymphocytes, 0.84% eosinophils, 0.17% basophils, and 7.6% monocytes. Liver function tests revealed elevated total bilirubin at 2.7 mg/dL, which increased to 10.58 mg/dL after five days; and AST of 42 U/L, which rose to 71 U/L. Coagulation studies were within normal limits (PT 13 seconds, INR 1.0). Inflammatory markers were elevated, with a C-reactive protein of 19 mg/dL. Renal function was significantly impaired, with creatinine 7 mg/dL, urea 145 mg/dL, potassium 5.4 mEq/L, sodium 135 mEq/L, and chloride 103 mEq/L. Blood cultures obtained were negative. Results for Rickettsiosis IgM (IFI), Hantavirus PCR, Dengue NS1, Dengue IgM, and rapid malaria testing were negative, with a positive result for Dengue IgG. Imaging: Chest imaging showed bilateral diffuse alveolar infiltrates on chest X-ray (Image B). A subsequent chest CT revealed extensive bilateral lung consolidation consistent with severe diffuse pneumonia, along with a tree-in-bud pattern indicating bronchiolar involvement. Additionally, pneumomediastinum with related subcutaneous emphysema was observed (Images C and D). UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
  Discussion: During hospitalization, a reactive microscopic agglutination test (MAT) showed titers of 1:400 for Hurstbridge, 1:800 for Icterohaemorrhagiae, and 1:400 for Varillal, along with positive Leptospira IgM antibodies. Leptospirosis is a zoonotic disease caused by a spirochete, most often Leptospira interrogans. It mainly affects rural farmers, people who work with livestock, individuals exposed to rodents, and populations living in impoverished urban or semi-urban areas. Infections can also happen through recreational activities involving contaminated water (1,2). It has a higher incidence in tropical and subtropical regions, including Southeast Asia, Sub-Saharan Africa, the Caribbean, and Oceania, and is strongly associated with flooding and poor sanitation (2). In Peru, the highest prevalence is seen in areas of the jungle, primarily Loreto, followed by Ucayali and San Martín; and some areas of the coast, such as Piura and Lima. So far this year, during the first three months, 1570 leptospirosis cases were reported, along with 12 deaths (3). Leptospirosis can often be underdiagnosed because its symptoms are nonspecific and resemble other febrile illnesses. Differential diagnoses include malaria, dengue, hepatitis, and yellow fever. It has a wide range of presentations, from asymptomatic infection to severe, life-threatening disease. In mild cases, it includes fever, headache, myalgias—particularly in the calves—conjunctival suffusion, nausea, and vomiting. However, the disease can progress to severe forms like Weil’s disease, which may present with jaundice, kidney failure, liver damage, bleeding, and complications such as meningitis or severe pulmonary hemorrhage (1,4). Diagnosis is based on clinical suspicion supported by laboratory tests; the gold standard is the Microagglutination test (MAT). Other available tests include IgM, which becomes detectable during the first week of disease; ELISA; dark field microscopy, often associated with poor sensitivity and specificity because it requires a high burden for adequate detection; PCR on samples such as blood, urine, or CSF; and culture (4,5). Management begins with early recognition and prompt antibiotic therapy. Mild cases can be, in some instances, self-limited and are usually treated with oral antibiotics, especially doxycycline, with alternatives such as azithromycin and amoxicillin. Severe cases require hospitalization, treatment with intravenous antibiotics like ceftriaxone, and supportive care, although the benefit of antibiotics in late-stage disease with organ dysfunction remains uncertain (6,7). Prevention focuses on reducing exposure to contaminated water or environments through protective measures, sanitation, rodent control, and access to clean water (6). On the day of admission, the patient was admitted to the ICU, requiring invasive mechanical ventilation and emergency hemodialysis. He was started on ceftriaxone 2 g every 24 hours for five days and furosemide 20 mg every eight hours. He was then transferred to the tropical ward on a 2 L nasal cannula and is currently breathing without oxygen support. His current medications include losartan and bisoprolol to control episodes of elevated blood pressure during hospitalization. The patient showed significant improvement, evidenced clinically by no longer needing oxygen support, laboratory by improved renal function, with the latest creatinine level at 0.99 mg/dL, and radiologically by improvement on imaging studies (Images E and F). References |
 |
Gorgas Case 2026-8 |
 |
|
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by the 2026 Gorgas Course participants.
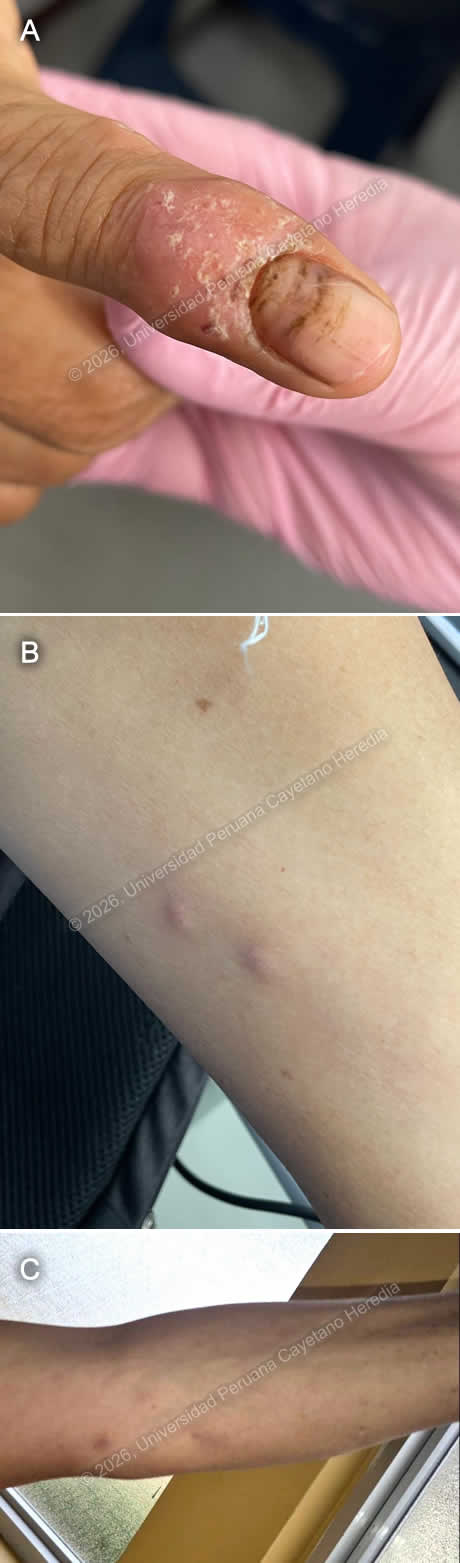
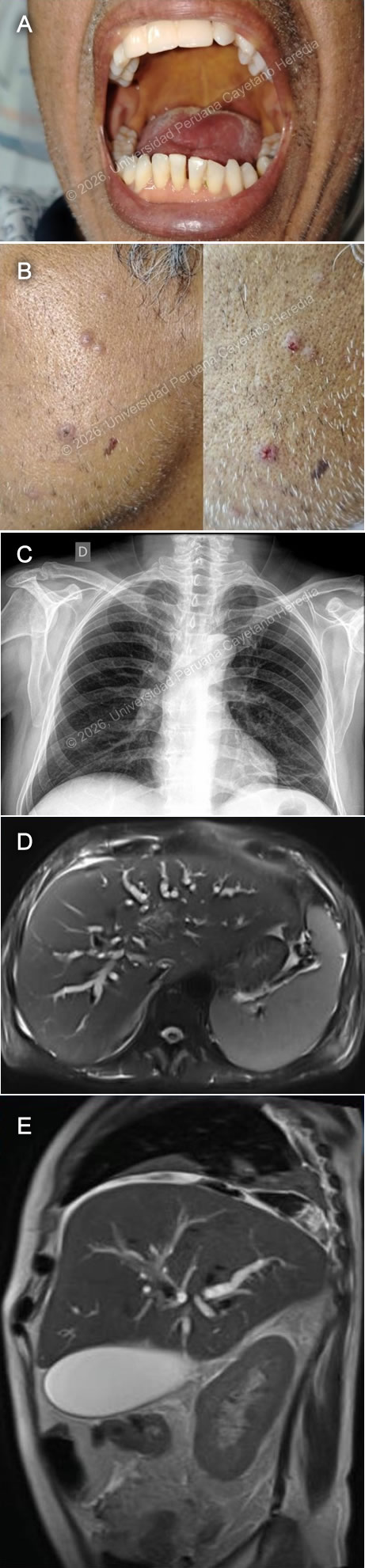
 History: The patient is a 41-year-old man from Lima who presents with a 6-month history of a progressive illness characterized by a pruritic maculopapular rash initially involving the arms and legs, which evolved into plaques with fine desquamation. This has been accompanied by intermittent fever and generalized malaise. Over the same period, he reports chronic watery diarrhea (5–6 episodes daily), including two episodes of bloody diarrhea, associated with anorexia, abdominal bloating, and unintentional weight loss of 14 kg. He also notes the development of whitish oral plaques and dysphagia to solid foods. He denies vomiting, seizures, loss of consciousness, cough, dyspnea, and night sweats. Epidemiology: The patient lives in Rimac, an urban area of Lima, where he works as a construction worker. He reports a history of alcohol use on weekends since adolescence, which he stopped 10 years ago. Currently, he smokes about one cigarette per week and uses marijuana. He also has a history of multiple female sexual partners (exact number unknown) with inconsistent condom use. Additionally, he previously had gonorrhea in 2010 and completed treatment for it. Physical examination: HR 76, RR 16, Temp 36.8°C, BP 120/70, SpO₂ 97% on room air. The patient appears with dry skin and mild pallor. Dermatologic examination shows plaque-like lesions mainly on the neck, arms, and legs, as well as hyperpigmented, scaly patches on the abdomen, back, palms, and soles (Images A,B,C,D). There is significant scaling of the scalp and eyebrows. Hair is sparse and brittle, with areas of alopecia (Image E). Palpation reveals non-tender lymphadenopathy in both inguinal regions. Genital exam shows erosive lesions on the penis with mild swelling and tenderness, along with whitish discharge at the balanopreputial sulcus; no ulcers are present. Neurological assessment indicates the patient is alert and oriented, with no focal deficits or meningeal signs. The remaining exam was unremarkable. Laboratory: Initial tests showed hemoglobin at 8.1 g/dL and leukocytes at 2500, with 64% neutrophils, 13.2% lymphocytes, 0.5% basophils, 10.4% monocytes, and 11.9% eosinophils. Platelet count was 291000. Total bilirubin was 0.5 mg/dL, alkaline phosphatase was 104, GGT was 35, and electrolytes and renal function were normal. An HIV test was positive, CD4 count was 35, viral load was 267000, and the HTLV-1 test was positive. A serum CrAg was positive. TB LAM was negative. Sputum GeneXpert Ultra was negative. A lumbar puncture was performed, revealing clear fluid with an opening pressure of 16 cmH2O. CSF analysis showed 2 leukocytes, 1 RBC, glucose at 46, capillary glucose at 180, and protein at 74. No organisms were seen on Gram stain, GeneXpert Ultra from the CSF was negative, LFA for cryptococcosis was negative in both blood and CSF, and Indian ink was negative. VDRL in CSF was negative. UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
  Discussion: The initial RPR was negative, but repeat testing after a prozone effect showed 1:512 dilutions. A skin punch biopsy from lesions on the right arm shows that the stratum corneum exhibits signs of parakeratosis. In contrast, the epidermis displays moderate acanthosis with lymphocyte exocytosis and occasional apoptotic keratinocytes (Image F). The dermis reveals a moderate inflammatory infiltrate composed of lymphocytes and plasma cells (Image G), distributed around skin appendages and involving both superficial and deep layers, along with some extravasated red blood cells and dilated blood vessels. Overall, these histological findings, when correlated with the clinical context, are consistent with secondary syphilis. Dark-field microscopy was not available. Syphilis is a preventable and curable infection caused by Treponema pallidum, transmitted mainly through sexual contact, but also vertically and via blood products, with increasing incidence worldwide (1). The latest data from the WHO indicates that an estimated 8 million adults aged 15 to 49 contracted syphilis in 2022. Co-infection between syphilis and HIV is common, as they share transmission routes and risk factors (2). HIV and HTLV are closely related retroviruses with similar structures, target cells, and transmission routes. As a result, coinfection is common in endemic regions; studies in the Peruvian Amazon have reported up to 10% prevalence of HIV and HTLV-1/2 coinfection (3). This is clinically significant because the interaction between these viruses can influence disease progression; patients can develop AIDS more rapidly and face higher mortality rates. HTLV-1 is also associated with immune system disruption, affecting the normal function of CD4+ and CD8+ T cells, which can weaken immune defenses and increase vulnerability to opportunistic infections (4). Syphilis presents with primary, secondary, latent, and tertiary stages. Secondary syphilis is the systemic phase that usually develops after hematogenous and lymphatic spread of the organism from the initial site of infection. It can occur weeks to a few months after the primary stage and may involve multiple organ systems. It is also highly infectious (2,5). Secondary syphilis is known for its wide range of clinical presentations, earning it the nickname the “great imitator.” It is characterized by diffuse skin lesions, often involving the palms and soles. Its presentation can vary in patients with HIV and may include other forms such as plaques, nodular lesions, painful target lesions, and pustular ulcers. Mucocutaneous lesions and lymphadenopathy may also be present. Additional manifestations can include patchy alopecia and, less commonly, organ involvement and systemic symptoms (6,7). Diagnosis is primarily based on serologic testing guided by clinical suspicion. Nontreponemal tests are used for screening and monitoring, with positive results confirmed by treponemal-specific assays. Direct detection methods such as dark-field microscopy or PCR may be used in select cases but are not routine practice (7). Treatment of choice for secondary syphilis is a single intramuscular dose of benzathine penicillin G. For patients with a penicillin allergy, alternatives such as doxycycline or ceftriaxone may be considered. After therapy, patients should be observed for the Jarisch–Herxheimer reaction. Follow-up with serial nontreponemal titers is recommended to ensure an adequate response to treatment. Tests are recommended at 3, 6, and, when indicated, 12 months post-treatment. A fourfold reduction in RPR titer or conversion to a non-reactive result indicates successful therapy (7,8). Our patient received treatment, under suspicion of possible neurologic involvement, with intravenous sodium penicillin for fourteen days, followed by a single intramuscular dose of penicillin G, with no adverse events. He also received treatment with fluconazole because of a positive serum cryptococcal antigen. And was started on prophylaxis with trimetoprim-sulfametoxazole. After ruling out other pathologies, he was started on antiretroviral therapy. References |
 |
Gorgas Case 2026-7 |
 |
|
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by the 2026 Gorgas Course participants.
 History: A 49-year-old woman presents with a six-month history of progressively enlarging nodular skin lesions. About one month after sustaining a wound on her finger while preparing fish, she noticed a non-painful, non-pruritic spot and a nodular lesion at the injury site. Over the following months, the lesion persisted, and additional nodular lesions appeared, extending along a lymphatic tract from the original site. She denies fever or other systemic symptoms. Epidemiology: The patient was born and currently lives in a rural area of the Peruvian highlands, in San Ignacio, Cajamarca, at about 1,324 meters above sea level. She works in agriculture and gardening on her own farm, which involves frequent contact with soil and plant material. She denies close contact with people who have similar lesions. Physical examination: HR: 74, RR: 18, T: 36.7°C, BP: 120/70, SatO2: 98% on room air. On physical exam, a primary lesion is seen on the finger at the initial injury site (Image A). Additional nodular lesions are present along the lymphatic vessels extending proximally from the primary lesion (Images B, C). They are not associated with warmth, pruritus, or tenderness. The rest of the physical exam is unremarkable and noncontributory.. Laboratory: Initial tests revealed hemoglobin at 15.1 g/dL and leukocytes at 4160, with 52.1% neutrophils, 39.7% lymphocytes, 0.2% basophils, 6.3% monocytes, and 1.7% eosinophils. Platelet count was 158000. Liver function tests revealed AST at 27.5 U/L (normal range 8 to 48 U/L) and ALT at 18.60 U/L (Normal range 7 to 55 U/L). Total bilirubin was 0.46 mg/dL, with direct bilirubin at 0.19. UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
 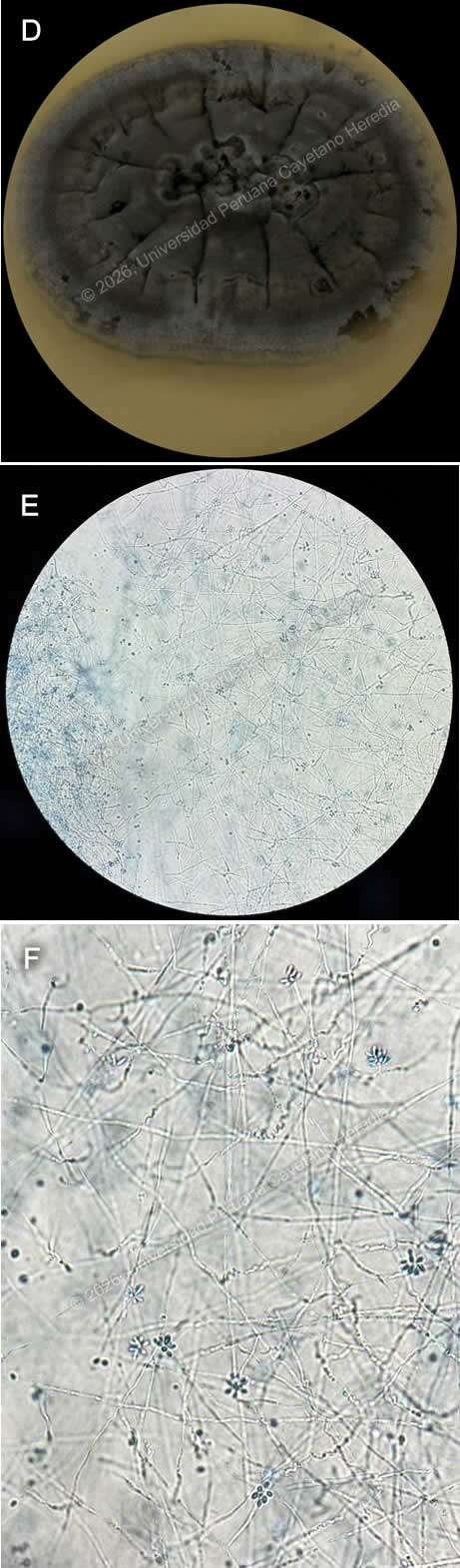 Discussion: A biopsy from the lesion was taken, direct observation was negative for Leishmania, blood agar culture for Leishmaniasis was negative as well. Culture for common germs and MGIT for Mycobacteria were negative. Agar Sabouraud fungal culture was positive for Sporothrix spp., confirming the diagnosis (Image D). Direct microscopic observation from the culture revealed thin hyphae, from which small, oval conidia arise, and often cluster in a “rosette” or “daisy-like” pattern, a feature suggestive of the genus Sporothrix (Images E, F). Sporotrichosis is a subcutaneous mycosis caused by dimorphic fungi of the genus Sporothrix, most commonly Sporothrix schenckii. It is found worldwide but is most prevalent in tropical and subtropical regions. It is frequently identified in Central and South America, the southern United States, Africa, and Southeast Asia, with recognized hyperendemic areas such as Guatemala, Mexico, Peru, and South Africa (1). In Peru, highland regions are considered endemic, with most cases reported from Abancay and Cajamarca (2). It is frequently associated with farming and gardening activities, existing as its mycelial or filamentous form in soil, wood, plant debris, and organic matter at lower temperatures around 25°C. In the human body, it transforms into yeast. Infection occurs when the organism is introduced into the skin through traumatic inoculation of the fungi (3). Clinical presentation may vary; it most frequently manifests as fixed cutaneous or lymphocutaneous, beginning with a lesion at the site of inoculation, followed by secondary lesions along lymphatic tracts. Rarely does it spread via the hematogenous route, leading to the disseminated form, which may present with organ involvement and multiple skin lesions at noncontiguous sites, and is associated with immunocompromised hosts (3,4). Diagnosis can be challenging because its clinical manifestations may resemble other conditions, such as cutaneous leishmaniasis, nocardiosis, tularemia, infections by atypical mycobacteria, and cutaneous tuberculosis. Therefore, diagnosis relies on a combination of clinical history, exposure, epidemiological context, and physical examination findings; it is supported by laboratory confirmation, with fungal culture being the gold standard. Other methods include direct microscopy, histopathology, and PCR to assist in identifying species (5,6). Management varies according to the clinical presentation and severity of the disease. Oral itraconazole is the treatment of choice for cutaneous and lymphocutaneous sporotrichosis and is typically continued for one month after complete clinical resolution is achieved. In more severe or disseminated forms of the infection, initial therapy is indicated with amphotericin B, followed by oral itraconazole (4,5). Our patient had the lymphocutaneous presentation and received Itraconazole; she is currently in her third month of treatment with no adverse events. References |
 |
Gorgas Case 2026-6 |
 |
|
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by the 2026 Gorgas Course participants.
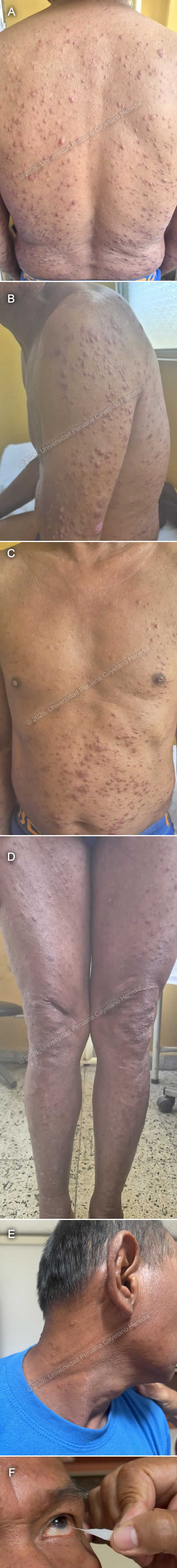 History: A 54-year-old man presents with a six-month history of progressively worsening generalized pruritic skin lesions that initially appeared as hives and papules, affecting the abdomen and upper limbs before spreading to the rest of the body. Three months later, he began experiencing sporadic nosebleeds and increasing numbness in his hands and feet. Due to decreased sensation in his hands, he has developed wounds from unnoticed trauma. In the last month, as his difficulty performing work-related activities worsened, he was advised to seek further medical evaluation. Over the past six months, the patient reports a weight loss of approximately 4 kg without trying. Epidemiology: The patient was born in a rural area of Yurimaguas, in the low jungle of Peru, and has been living in Lima for the past 16 years. He is currently a warehouse worker in Lima. When he was in the jungle, he worked as a farmer for 6 years, cultivating corn, rice, bananas, and cassava, and raising animals including pigs, chickens, and cattle. He reports a past history of cutaneous leishmaniasis at age 12, with complete antimonial treatment. He also mentions having a nasal septum perforation since he was 18, with no clear cause. He denies sick contacts. Physical examination: His vital signs were HR: 78 beats per minute, RR: 18 breaths per minute, BP: 110/70 mmHg, T: 36.7°C, SatO2: 98% on room air. Skin examination revealed multiple very superficial papules and nodules in a dispersed pattern involving his face, abdomen, back, and both upper and lower limbs (Images A, B, C, D). These were associated with nerve thickening (Image E), corneal anesthesia with absence of corneal reflex (Image F), and decreased sensation in the hands and feet. A perforated septum was also observed. The rest of the exam was unremarkable. Laboratory: Initial tests showed hemoglobin at 13.2 g/dL and leukocytes at 5530, with 68.9% neutrophils, 22.2% lymphocytes, 0.2% basophils, 7.6% monocytes, and 1.1% eosinophils. Platelet count was 437000. Liver function tests revealed AST at 17 U/L (normal range 8 to 48 U/L) and ALT at 10 U/L (Normal range 7 to 55 U/L). Total bilirubin was 0.75, with direct bilirubin at 0.3. Renal function was within normal limits, with urea at 21 mg/dL and creatinine at 1.02 mg/dL. Glucose was 68 mg/dL. UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
  Discussion: A skin punch biopsy from the lesions on the back was performed, revealing a moderate inflammatory infiltrate in the dermis with a nodular and linear pattern. It consisted of vacuolated histiocytes containing multiple bacillary structures. Fite–Faraco staining (Image G) is positive (3+), confirming the diagnosis of lepromatous leprosy. Leprosy is a chronic granulomatous infection caused by Mycobacterium leprae. It remains a neglected tropical disease present in over 120 countries, with approximately 200,000 new cases each year. Although it was declared eliminated as a global public health problem by 2000, new cases still appear in countries like Brazil, India, and Indonesia, while many nations now report few or no cases (1). In Peru, leprosy is endemic in Amazon regions, especially in the departments of Ucayali, Loreto, Amazonas, Huánuco, and San Martín (2). Twenty six new cases were reported in 2023, all of them from the departments of Loreto and Ucayali (3). Transmission is unclear, though it is thought to occur via respiratory droplets from the nose and mouth of untreated individuals during prolonged close contact (1,4). The incubation period is variable, from months to years, and it commonly affects peripheral nerves, skin, and mucosa (4,5). And if left untreated, it can lead to severe disability, mostly secondary to impairment of nerve function or visible deformity. Clinical manifestations vary among patients due to differences in individual immune responses, and they are classified into Tuberculoid (TT), Lepromatous (LL), Borderline Lepromatous (BL), Mid Borderline (BB), and Borderline Tuberculoid (BT) (6). Lepromatous leprosy is linked to a Th2-dominant immune response to the pathogen and typically presents with widespread, diffuse infiltration of the skin and very superficial papules and nodules (lepromas) containing numerous organisms, along with nerve involvement (5). Diagnosis is made through clinical history and examination, with slit-skin smear microscopy or biopsy with histopathology as needed. Diagnosis requires at least one of the following criteria: sensory loss in skin lesions, thickened peripheral nerves, or detection of acid-fast bacilli on slit-skin smears (7). The absence of reliable point-of-care tests often contributes to delayed diagnosis and increased risk of nerve damage and disability (1,5). WHO guidelines for treatment recommend a three-drug regimen with rifampicin, dapsone, and clofazimine with variable duration depending on the clinical category of presentation, with a recommended 12-month regimen for multibacillary leprosy (1). Monthly RMM is the standard in the US and in countries where clofazimine cannot be obtained (8). Our patient has multibacillary lepromatous leprosy and was started on rifampicin, dapsone, and clofazimine. He is currently undergoing his second month of treatment with no adverse events (Images H, I). Complications can persist even after bacteriological cure and include immune reactions, nerve damage, ocular sequelae, and disabilities. Management is not limited to medication, it also needs long-term support to address the physical, psychological, and social impacts of the disease (9). We would like to thank Dr. David Freedman for his contributions. References |
 |
Gorgas Case 2026-5 |
 |
|
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by the 2026 Gorgas Course participants.
 History: A 25-year-old woman from the highlands of Huancavelica presented with a 4-year history of gradually worsening abdominal pain. Initially, she experienced mild, diffuse upper abdominal pain after meals, which was later accompanied by nausea, decreased appetite, and postprandial vomiting (≥2 episodes daily). Nine days before admission, her condition worsened with increased fatigue and general malaise. Due to persistent symptoms, she sought hospital care and was admitted for further evaluation and management. On review of systems, she describes mild to moderate global headaches and subjective fevers but denies visual disturbances or a history of seizures. Epidemiology: The patient was born with an imperforate anus and underwent stoma surgery at 1 year old at Cayetano Heredia Hospital. She is originally from and currently lives in Izcuchaca, Huancavelica, a rural highland area in Peru, at 2,900 meters above sea level. She sells food on the streets and has incomplete primary education. She lives with relatives in a household without access to potable water, and reports multiple exposures to farm animals (hens, cows, guinea pigs, pigs) and currently owns sheep. She denies exposure to vectors, travel history, and sick contacts. Physical examination: HR: 81 bpm, RR: 20 breaths per minute, T: 36.5°C, BP: 100/70, SpO2: 97% on room air. On abdominal examination, a stoma was noted in the epigastric region, with tenderness on palpation of the upper abdomen. The rest of the physical exam was unremarkable. An imperforate anus was also present. Laboratory: Initial laboratory tests showed a normal hemoglobin of 12.2 and leukocytes of 4400; with 53.4% neutrophils, 36.6% lymphocytes, 0.3% basophils, 4.9% monocytes, and 4.8% eosinophils. Platelet count was 221,000. Liver function tests revealed AST 79 U/L (normal value 8 to 48 U/L) and ALT 65 U/L (Normal range 7 to 55 U/L). Alkaline phosphatase of 25 U/L (normal range 30 to 130 U/L), and GGT of 184 (normal range 5 to 40 U/L). Total bilirubin was 0.3, with direct bilirubin 0.2. Renal function was within normal limits, with urea 27 mg/dL and creatinine 0.7 mg/dL. Electrolytes were also normal, including sodium 137 mEq/L, potassium 4.63 mEq/L, and chloride 102 mEq/L. Albumin was 4.4 g/dL. HIV and RPR were negative. Imaging: Upper and lower limb X-rays revealed multiple calcifications in the muscles (Images A, B), Abdominal CT showed hepatic nodules, ileocolic lymphadenopathy, and a distended ascendant colon with cystic lesions in the mesenteric adipose tissue (Image C), multiple cystic lesions are also evidenced in the pelvic MRI (Image D). Neuroimaging revealed multiple intracranial cystic and calcified lesions on head CT (Image E), and brain MRI confirmed multiple intraparenchymal cysts with ventricular involvement (Image F). UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
  Discussion: A serum Western Blot assay was positive for cysticercosis. Cysticercosis is a neglected tropical disease and a major global public health issue, most common in developing countries, especially in regions of Asia, sub-Saharan Africa, and Latin America (1). In Peru, the highest prevalence occurs in the highlands, particularly in Cuzco, Huancayo, and Andahuaylas; on the northern coast in Tumbes, and in some areas of the high jungle (2). The parasite has a two-host life cycle involving a definitive host and an intermediate host. Humans are the only definitive hosts for the adult tapeworm and can also serve as accidental intermediate hosts. Pigs are the typical intermediate hosts in endemic areas, harboring the larval form in their tissues (3). Cysticercosis is acquired through the fecal-oral route after the ingestion of Taenia solium eggs in contaminated food or water. After ingestion, the eggs hatch into oncospheres, which penetrate the intestinal wall and disseminate hematogenously to various tissues, where they develop into cysticerci and can localize in muscle, subcutaneous tissue, eyes, and particularly the central nervous system (1,3). The infection can stay asymptomatic throughout life, especially in endemic areas, or show a range of clinical signs depending on the location, size, number, and stage of the lesions (4). These features can also influence prognosis and treatment options. Diagnosis mainly relies on imaging studies, supported by serologic tests such as ELISA and Western Blot (5). Our patient presents with disseminated cysticercosis involving soft tissues, the intestine, and the brain. In this patient, NCC was characterized by inactive calcified lesions, active cystic lesions, and intraventricular cystic lesions. Clinical presentation of disseminated cysticercosis is rare and affects the brain, muscles, subcutaneous tissue, eyes, and, sometimes, organs such as the liver and heart. It is diagnosed when cysticerci are found in at least two separate organ systems, confirmed by imaging or histopathology. It has been associated with risk factors including residence in highly endemic areas, and altered immune response (7,8). Given the uncommon nature of this presentation, clear management guidelines have not yet been established (9). Our patient underwent a right colectomy with extended ileal resection, jejunum–transverse anastomosis, removal of mesenteric adipose tissue, and colostomy revision. Histopathological examination of the surgical specimen demonstrated segments of small and large intestine with multiple cystic lesions involving the submucosa of the small intestine and the mesentery, some of which were calcified. Parasitic structures consistent with cysticerci were identified in the mesenteric adipose tissue (Image G). She is currently awaiting endoscopic removal of the intraventricular cyst, which will be followed by antiparasitic treatment. References |
 |
Gorgas Case 2026-4 |
 |
|
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by the 2026 Gorgas Course participants.
 History: A 53-year-old woman presented with a 20-day history of progressively worsening dyspnea and cough. Her symptoms began with mild shortness of breath and a productive cough, accompanied by subjective fever, malaise, and night sweats. Over the following days, her dyspnea intensified, and the cough became blood tinged. On the day of admission, she developed dyspnea at rest, prompting her to seek care in the Emergency Department, where she was subsequently admitted. She reported intermittent abdominal pain over the past two years, unintentional weight loss of five kilograms in the last month, and pruritus of the hands and feet. Epidemiology: The patient is a native of Ancash (Congos–Ocras) and currently resides in Ocros. He works as an agricultural worker and livestock breeder, with frequent direct exposure to domestic animals, including cattle, donkeys, horses, poultry, pigs, sheep, and guinea pigs. He reports consuming home-produced dairy products and watercress since childhood. Physical examination: HR: 82 x’, RR: 22 x’, Temp: 36.6°C, BP: 100/70, SatO2: 92% on room air. On admission, the patient was afebrile, hemodynamically stable, and had low oxygen saturation on room air. Skin examination revealed mild pallor. Respiratory examination showed tachypnea and bilateral wheezing. The remainder of the physical examination was unremarkable. Laboratory: Initial laboratory tests showed a normal hemoglobin of 14.7 and leukocytosis of 36,300; with 15.9% neutrophils, 4% lymphocytes, 1% basophils, 1% monocytes, and marked eosinophilia at 74.4%, corresponding to an absolute eosinophil count of 27,007. Platelet count was 281,000. Liver function tests revealed elevated enzymes; AST 78 U/L (normal value 8 to 48 U/L) and ALT 199 U/L (Normal range 7 to 55 U/L). There was also a significant increase in alkaline phosphatase to 609 (normal range 30 to 130 U/L), and GGT of 143 (normal range 5 to 40 U/L). Total bilirubin was 0.8, with direct bilirubin 0.4. Renal function was within normal limits, with urea 40 mg/dL and creatinine 1 mg/dL. Electrolytes were also normal, including sodium 140 mEq/L, potassium 4.94 mEq/L, and chloride 98 mEq/L. Total protein was 9 g/dL, and albumin was 4.4 g/dL. A five-method stool examination was positive for Blastocystis sp. and Entamoeba coli. Imaging: A chest X-ray shows a reticular pattern in both lung fields (Image A), and a Chest CT scan shows ground-glass infiltrates and fibrotic bands in the bases of both lung fields (Image B). An abdominal CT scan that shows hepatomegaly and the presence of hypodense lesions in the liver parenchyma (Image C). UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
  Discussion: A FAS-2 ELISA test was performed, which was positive, confirming the diagnosis of fascioliasis. Fascioliasis is a zoonotic, food-borne parasitic disease caused by the trematodes Fasciola hepatica and F. gigantica, particularly in Africa and Asia. Also known as “liver flukes,” it infects humans as well as a variety of domestic and wild animal hosts. It is acquired by ingesting encysted metacercariae attached to aquatic vegetation, most commonly watercress, in stagnant or slow-moving freshwater environments (1). The highest prevalence is reported in areas where livestock farming is common and raw aquatic plants are frequently consumed, such as South America, Africa, and Asia (2). In the Peruvian highlands, the highest rates are observed in Cajamarca, Cusco, Puno, Junín, and Ancash (3). Clinical presentation depends on the phase of infection. The acute phase is caused by larval migration through the intestine and liver, often involving tissue damage and inflammation; it can present with fever, abdominal pain, and eosinophilia. The chronic phase involves adult flukes in the bile ducts, in some cases for years, causing inflammation and obstruction, which can lead to complications such as cholangitis, cholecystitis, gallstones, liver abscesses, or cirrhosis (4). In acute fascioliasis, eosinophilia occurs in nearly all cases. Our patient exhibited hypereosinophilia and progressively worsening respiratory symptoms consistent with acute eosinophilic pneumonia, as a rare complication previously reported (5), in which eosinophilic infiltration and degranulation lead to lung inflammation and the associated clinical manifestations (6). Other possible laboratory findings include anemia, commonly due to chronic biliary blood loss, and liver enzymes and bilirubin levels are frequently elevated during both larval migration and the chronic stage. For diagnostic confirmation, serology is more sensitive than stool testing, especially in non-endemic regions. Our patient had a positive serology for FAS-2 ELISA and two negative stool Kato-Katz samples. Management of acute eosinophilic pneumonia includes high-dose steroids, which typically improve symptoms within 48 hours (6). Along with addressing the underlying cause. Triclabendazole is the treatment of choice for fascioliasis, with high cure rates and an acceptable safety profile. Although mass treatment has been proposed for control in humans and livestock, inconsistent administration, particularly in resource-limited settings, may promote the development of drug resistance (2). Our patient was treated with triclabendazole 750 mg daily for two days along with corticosteroids. Despite initial worsening of respiratory distress, also evidenced in imaging (Image D), there was marked improvement within 48 hours after starting prednisone at 1 mg/kg per day for one week. Following therapy, the patient showed significant laboratory and rapid clinical recovery, with complete resolution of respiratory failure and eosinophilia. References |
 |
Gorgas Case 2026-3 |
 |
|
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by participants in the 2026 Gorgas Course.
 History: A 54-year-old man presented with a 3-month history of abdominal pain, weight loss, fever, and jaundice. Symptoms began with nausea and scleral/mucosal jaundice, and one month later, fatigue, poor appetite, postprandial epigastric pain, and bloating. He later developed pale stools, dark urine, night sweats, and fevers up to 38 °C. During the third month, he was empirically treated at a GI clinic with steroids (prednisone 40 mg/qd for one week) for a presumptive diagnosis of IgG4 disease. A week and a half later, he developed generalized lymphadenopathy and a papular skin rash. Worsening symptoms prompted an ED presentation and hospital admission for further evaluation. Epidemiology: The patient has a history of cutaneous leishmaniasis diagnosed and treated in 2022, which recurred in 2023 and healed with residual scarring. He was born in Yurumarca, Chachapoyas, in the high jungle of Peru. In adolescence, he moved to Lima. From 2020 to 2024, he lived and worked on a farm in Vituya, in the high jungle, where he cultivated dragon fruit, lemons, bananas, and oranges, and harvested coffee. He frequently swam in nearby rivers, visited caves while resting from farm work, raised multiple domestic animals, including guinea pigs, cattle, chickens, pigs, and donkeys, and reported exposure to rodents and bats. He consumed home-produced foods, including unpasteurized dairy products, and experienced frequent insect and mosquito bites. He denied known sick contacts, though neighbors reportedly had untreated skin lesions compatible with leishmaniasis. His home lacked electricity and potable water. Physical examination: HR: 103x’, RR: 21x’, BP: 120/80, Temp: 38°C, SatO2: 95% on room air. Clinical examination revealed jaundice of the skin, sclerae, and mucous membranes, along with pallor (Image A). Papular and crusted skin lesions were present on the face, submental region, and upper and lower extremities (Image B). An atrophic, hypopigmented scar was noted on the posterior left upper extremity. Oral examination revealed a full dentition without thrush or ulcers. Bilateral lymphadenopathy was noted, predominantly tender and soft, affecting the retroauricular, submandibular/submaxillary, axillary, and inguinal regions, with most nodes mobile and dry. The abdomen was distended, with right upper quadrant tenderness on palpation. The liver was palpable 3 cm below the right costal margin, and splenomegaly was noted. The rest of the exam was unremarkable. Laboratory: Basic analysis showed mild normocytic normochromic anemia with hemoglobin in 10.5 g/dL, with 8700 leukocytes (86% neutrophils, 8.6% lymphocytes, 1% eosinophils, 0.1% basophils, 4.3% monocytes). Glucose was 79 mg/dL. Renal function remained preserved (creatinine 0.7 mg/dL, urea 29–25 mg/dL), with normal electrolytes. Liver function tests were increased, with alkaline phosphatase of 920 U/L (Normal range: 40 to 129 U/L) and gamma-glutamyl transferase 1142 U/L (Normal range: 5-40 U/L), associated with total bilirubin of 17.4 mg/dL (Normal range: 0.1 to 1.2 mg/dL), with direct predominance (15 mg/dL). Liver transaminases were elevated AST/ALT: 119/120. Coagulation studies showed prolonged PT 25.6 and INR 2.07 and PTT 62. Lipid profile revealed hypercholesterolemia 219 mg/dL and hypertriglyceridemia 224 mg/dL. Amylase was 50 U/L (normal range: 30–140 U/L), lipase was 23 U/L (normal range: 0 to 160 U/L). Imaging: A chest X ray revealed mild bilateral accentuation of the bronchovascular markings, with no other significant findings (Image C), a magnetic resonance cholangiography showed marked dilation of the intrahepatic bile ducts secondary to diffuse mural thickening of the extrahepatic bile ducts, along with signs of hepatosplenomegaly, retroperitoneal lymphadenopathy, and gallbladder hydrops with minimal biliary sludge (Images D and E). UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
  Discussion: KOH examination of an induced sputum sample (Image F) revealed large, oval, double-walled yeast cells with multiple budding daughter cells, characteristic of Paracoccidioides species. Our patient underwent ERCP, during which a biliary sphincterotomy with balloon sweep was performed, and a biliary stent was placed. Nodular erosive lesions were observed in the duodenum, and a biopsy was obtained. Histopathologic evaluation of duodenal mucosa reported glandular architectural changes and moderate chronic inflammation with the presence of yeasts; no granulomas were reported (Image G), and a lymph node biopsy (Image H) demonstrated a mixed necrotizing granulomatous inflammatory process with abundant yeast-like fungal structures of variable size on hematoxylin and eosin staining. Paracoccidioidomycosis is an endemic mycosis caused by fungi of the genus Paracoccidioides, predominantly Paracoccidioides brasiliensis. It primarily affects rural populations in Latin America, particularly in tropical regions such as Brazil, Peru, Venezuela, Ecuador, and Colombia (1). It is strongly associated with agricultural work, where exposure to soil is important. Paracoccidioides species reside in the environment in their mycelial form and are acquired through inhalation; once inside the host, the fungus undergoes thermal dimorphism, transforming into its yeast form (2). Paracoccidioidomycosis presents with diverse clinical manifestations and is generally categorized into two main forms: the acute/subacute (juvenile) form and the chronic (adult) form. In severely immunocompromised patients, it may present with features of both forms (3). The juvenile form is the least common, occurring in 5% to 25% of cases, and it primarily affects children and young adults under 35 years, with equal gender distribution. It progresses rapidly, typically presenting with fever, weight loss, and generalized lymphadenopathy. Hepatosplenomegaly, abdominal symptoms, skin lesions, and, occasionally, bone involvement may occur, while pulmonary involvement is uncommon (2,3). The chronic form accounts for 75% to 95% of cases and typically affects men aged 30 to 60 years. It has a slow onset and may be localized or disseminated. Pulmonary involvement is most common, presenting with chronic respiratory symptoms and weight loss. Mucocutaneous oral lesions are frequent, and adrenal involvement may occur. Fever and significant lymphadenopathy are uncommon (2,3). Although our patient is outside the typical age range for the juvenile form, he meets clinical criteria consistent with this presentation. Additionally, high-dose corticosteroids may have contributed to accelerated clinical progression. Paracoccidioidomycosis is diagnosed by demonstrating P. brasiliensis in clinical specimens. The fungus can be identified by direct microscopy of sputum, skin scrapings, or biopsy material, including tissue sections stained with hematoxylin and eosin or Gomori–Grocott. Serologic assays for antibodies or antigens may support the diagnosis, although their sensitivity varies and cross-reactivity with other endemic mycoses can occur. Additional diagnostic tools include PCR techniques and fungal culture (4). All patients with the juvenile form should receive amphotericin B. The deoxycholate formulation is administered at 0.5–0.7 mg/kg/day (maximum 50 mg/day). Once clinical stabilization is achieved, the regimen is transitioned to an oral azole derivative or trimethoprim–sulfamethoxazole (3, 5). Our patient is currently receiving amphotericin B and is showing clinical and laboratory improvement, with a significant decrease in previously elevated liver function tests. References |
 |
Gorgas Case 2026-2 |
 |
|
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by the 2026 Gorgas Course participants.
 History: A 60-year-old male from Lima presented with a 6-year history of a progressively enlarging left nasal lesion and dysphagia. The lesion initially appeared as a painless papule with mild pruritus that gradually increased in size. By the fifth year of disease, the lesion became pustular after self-manipulation, with progressive growth and blood-tinged discharge. The nasal lesion became crusted and extended bilaterally, causing partial nasal obstruction, sleep-related breathing difficulty, nasal discharge, hoarseness, and dysphagia. In the last seven months, he noticed worsening of the symptoms. Due to the burden and progression of symptoms, he sought medical attention and was subsequently hospitalized. Epidemiology: The patient was born in Lima and currently resides in the district of Carabayllo, where he works as a construction worker. Travel history includes Aguaytia, a city in the high jungle of Peru, where he went thirty-two years ago and stayed for two years, working as a farmer harvesting bananas and cassava. In his second year, he developed a painless, ulcerative lesion with regular, elevated borders on the elbow. He didn’t seek medical care, and it self-limited in approximately three months, leaving an atrophic, hypochromic scar with irregular edges. He reports that during his stay, many of his co-workers presented similar lesions. Past medical history is notable for pulmonary tuberculosis, diagnosed seven months prior to admission after he developed cough, dyspnea, and weight loss. He completed treatment. Physical examination: HR: 81 x’, RR: 17 x’, BP: 102/66, Temp: 36.5, SatO2: 97%, FiO2: 0.21. The patient presents with marked nasal abnormalities, including an infiltrative, ulcerated lesion with irregular borders measuring approximately 3 × 3 cm in the left nasal cavity, associated with deformity, partial amputation of the left nostril, and a nasal septum perforation. Oral examination reveals a granulomatous lesion involving the hard palate and a nodular lesion at the apex of the tongue (Images A and B). Cutaneous findings include an atrophic, hypopigmented scar with irregular borders measuring approximately 6 × 2 cm on the elbow (Image C). A mobile left cervical lymph node measuring approximately 1 × 1 cm was also noted. The rest of the exam was unremarkable. Laboratory: Initial laboratory findings included hemoglobin of 9.9 g/dL, a white blood cell count of 5.8 ×10⁹/L with 68.8% neutrophils, and a platelet count of 576 ×10⁹/L. The metabolic panel was normal (urea 35 mg/dL, creatinine 0.5 mg/dL, sodium 137 mEq/L, potassium 4 mEq/L, chloride 95 mEq/L, and glucose of 88 mg/dL). Liver function tests and coagulation tests were also normal. Serologic testing for HIV was negative. A Montenegro skin test was positive (15x15mm). Imaging: A chest X-ray showed a nodular bilateral pattern, predominantly. Marked laryngeal thickening and narrowing. A neck lateral X ray showed thickening of the oropharyngeal region with visible obstruction (Image D). A CT scan showed similar findings along with cervical lymph nodes (Image E). UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
  Discussion: The patient was diagnosed with extensive mucosal leishmaniasis. Significant clinical findings were noted after fiberoptic bronchoscopy, revealing a congested pharynx, epiglottis, and larynx with multiple friable granulomatous lesions, edematous vocal cords, and approximately 10% patency of the surrounding mucosal opening (Image F). A nasal mucosal biopsy demonstrated inflammatory infiltrates with fibrosis and intracellular structures morphologically consistent with amastigotes (Image G), confirming the diagnosis of mucosal leishmaniasis. Leishmaniasis is a vector-borne parasitic disease caused by Leishmania species, transmitted by infected female sandflies (Phlebotomus and Lutzomyia). It is endemic in tropical and subtropical regions. The World Health Organization reports an estimated annual incidence of 700,000 to 1 million new leishmaniasis cases, with the majority occurring in the African region and the Americas (1). It is an endemic infectious disease in Peru, Leishmania (Viannia) braziliensis is the principal etiologic agent of mucocutaneous leishmaniasis, with L. (V.) guyanensis, L. (V.) peruviana, and rarely L. (L.) amazonensis also reported (2). With more than 4,000 cases reported annually, it is predominant in rural areas of the jungle and highlands. The highest incidence is observed in the departments of Madre de Dios, Cuzco, Loreto, Ucayali, and Huanuco (3). This disease manifests in three main clinical forms: cutaneous, mucocutaneous, and visceral, with the cutaneous form being the most frequent. The clinical expression and severity of the disease are influenced by parasite, host, and vector-related factors (4). Mucocutaneous leishmaniasis most commonly presents as a late sequela of the cutaneous form, although concurrent presentation may occur (1). Approximately 90% of cases are associated with a history of prior cutaneous lesions, often evidenced by residual scarring, as in our patient’s case (Image C). Mucocutaneous leishmaniasis most commonly presents with nasal involvement, and as the disease progresses, lesions may extend to the oral cavity, palate, pharynx, or larynx; presenting with erythema, edema, infiltration, nodular or ulcerative lesions, and, in advanced cases, destructive changes (2). Diagnosis is based on clinical manifestations and epidemiologic factors, and is confirmed by identification of the species through microscopic examination of lesion aspirates or biopsies, culture, and molecular methods (5). Differential diagnosis includes paracoccidioidomycosis, tuberculosis, histoplasmosis, syphilis, rhinoescleroma, granulomatosis with polyangiitis, or extranodal T-cell lymphoma. Hospitalization was required for systemic treatment and observation due to our patient’s severe clinical presentation. Amphotericin B deoxycholate 0.7-1 mg/kg IV is recommended for extensive mucosal compromise and laryngeal and pharyngeal involvement (5, 6, 7). Despite the lack of large, randomized trials to guide dosing or duration, adjunctive short-term steroids may be recommended for patients with severe inflammatory disease and airway-threatening edema, with tapering guided by clinical response (2). ENT evaluation is also advised to manage and follow airway involvement (5). Our patient is receiving systemic treatment with Amphotericin B deoxycholate with a target of a cumulative dose of 25 mg/kg, and a course of steroids with significant clinical improvement. References |
 |
Gorgas Case 2026-1 |
 |
|
The Gorgas Course in Clinical Tropical Medicine is held at the Institute of Tropical Medicine at Universidad Peruana Cayetano Heredia in Lima, Peru. Now in its 30th consecutive year, we are delighted to present a selection of intriguing cases encountered by course participants during the February/March sessions. The 9-week Gorgas Course in Clinical Tropical Medicine is currently underway. New cases will be distributed via email each week for the 9 weeks. Each case features a concise clinical history and relevant digital images, followed by the diagnosis and a brief discussion.
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by the 2026 Gorgas Course participants.  History: A 55-year-old male from the port of Callao, Lima, presented with a 2-day history of a painful vesiculobullous lesion on the right lower extremity. Symptoms began abruptly upon waking with sudden, intense pain in the posterior right thigh. Several hours later, he developed localized pruritus and burning and self-medicated with an unspecified antibiotic. The pain progressively worsened to severe intensity (10/10), was exacerbated by pressure, and was only minimally responsive to analgesics. One day prior to admission, he noted increased local warmth and persistent pain, followed by a violaceous bullous lesion and yellowish skin discoloration. On the day of admission, ongoing symptoms were accompanied by dark-colored urine, prompting presentation to the emergency department. He denies fever, prior skin trauma, nausea, vomiting, and generalized muscle cramps. Epidemiology: The patient had been temporarily working as a miner in Nazca and was residing in a rudimentary wooden structure without basic services or flooring, with reported exposure to rodents and spiders. Physical Examination on admission: HR: 100x’, RR: 20x’, Temperature: 37°C, BP: 104/71 mmHg, SatO2: 97% on room air. Skin examination revealed a confluent violaceous plaque measuring approximately 8 × 8 cm with associated violaceous bullae on the affected extremity (Image A,B), and jaundice. The patient was alert, well appearing, and in no acute distress. There was no peripheral edema or lymphadenopathy. Cardiopulmonary examination was unremarkable with clear lung fields and regular heart rate and rhythm. The abdomen was soft and non-tender, without hepatosplenomegaly. There was no costovertebral angle tenderness. Neurologic examination showed no focal deficits, and there were no meningeal signs. Laboratory: Initial laboratory evaluation was notable for leukocytosis with neutrophilia (WBC 11.9 ×10⁹/L, ANC 9.77), marked azotemia (creatinine 4.2 mg/dL, urea 136 mg/dL), a decline in hemoglobin from 12.1 g/dL to 8.8 g/dL was observed. And predominantly indirect hyperbilirubinemia (total 8.3 mg/dL; direct 1.5 mg/dL; indirect 6.8 mg/dL), consistent with hemolysis, along with AST-predominant transaminitis (AST 184 U/L, ALT 29 U/L), GGT was 35 U/L, and alkaline phosphatase was 71 U/L. There were marked elevations of LDH and CPK (2418 U/L and 673 U/L, respectively). PT was 15.3, INR 1.17 and PTT was 26.9. Urinalysis showed 3+ bacteria and granular casts, and arterial blood gas revealed respiratory alkalosis (pH 7.47, PCO₂ 27.1 mmHg). UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
  Discussion: Although the patient did not identify the culprit spider, he had noticed several brown spiders in his cabinet. The history and clinical presentation in this case are typical of loxoscelism, which is caused by envenomation from a Loxosceles species bite. These are cosmopolitan arachnids, with over 140 species described worldwide. The species of greatest clinical relevance in South America are L. intermedia, L. laeta, and L. gaucho, commonly known as violin spiders for the violin-shaped marking on the cephalothorax (1). However, the violin mark is not distinctive of the genus Loxosceles; correct identification includes the disposition of its six eyes and the lack of spines on its legs (2), Image C. Spider bites occur most frequently during warmer months, particularly spring and summer. Loxosceles bites usually occur at night in dry, dark indoor environments. The pathophysiology of the disease relies on the venom component, sphingomyelinase D, that causes endothelial damage, activates the complement system, and exerts direct cytotoxic effects that amplify the inflammatory response, leading to severe local or systemic reactions (1). A recent analysis of the components of the Peruvian L. laeta indicates that 69% of the venom contains Phospholipase-D, followed by metalloproteases (20,72 %), sicaritoxins (6,03 %), serine-proteases (2,28 %), hyaluronidases (1,80 %), and Translationally Controlled Tumor Protein (TCTP) (0,56 %) (3). During 2024, the Ministry of Health in Peru reported 1315 cases, with a cumulative number of more than 10,000 in the last 10 years. The jungle of the country concentrated most of the cases, followed by Lima (4). There are two clinical forms: cutaneous and systemic. The first is the most frequent and least severe, characterized by local and regional findings at the bite site, usually associated with skin necrosis appearing as an irregular area of ecchymosis or a livedoid plaque, along with erythema, edema, pain, and pruritus (1,5). The less frequently seen clinical manifestation is the viscerocutaneous or systemic presentation; it is potentially life-threatening and can present with fever, jaundice, disseminated intravascular coagulation, hemolysis, thrombocytopenia, and acute renal failure. Systemic symptoms can appear within 24 to 48 hours and are usually nonspecific (5,6). Loxoscelism is diagnosed clinically by integrating epidemiologic factors, clinical history, and characteristic cutaneous manifestations. Laboratory evaluation may be useful in identifying complications associated with systemic or viscerocutaneous presentation (7,8). Differential diagnosis includes accidents caused by other venomous animals, such as scorpions, snakes, or other spiders; vasculitis; hematologic or coagulation disorders, for example, hemolytic uremic syndrome and autoimmune hemolytic anemia; and other infectious causes, including ecthyma gangrenosum, cutaneous anthrax, or toxic shock syndrome (1)/. Management is primarily supportive and includes local wound care and symptomatic treatment with analgesics and antihistamines. Hospitalization is indicated for patients with systemic manifestations or rapidly progressive lesions. Our patient received one vial of heterologous antivenom and analgesics. Data on the use of antivenom or other medications for Loxosceles bites are conflicting; recommendations may vary. No therapy has demonstrated benefit beyond supportive care, and no medications have been shown to shorten healing time (1,4). Administration of specific anti-Loxosceles antivenom might be recommended, preferably within the first 24 hours or as soon as it is recognized; it is not advised after 72 hours (4,9). In other regions where the antivenom may not be available, short courses of systemic steroids are frequently used (4,7). The patient significantly improved; hemolysis stopped by the third day of hospitalization, but renal damage was established. A non-oliguric form of AKI remains, with no need for dialysis. Plastic surgeons have evaluated the patient for further intervention. References |