| 2004 Case #9 |  |
|
| Diagnosis: Leptospirosis. |
|
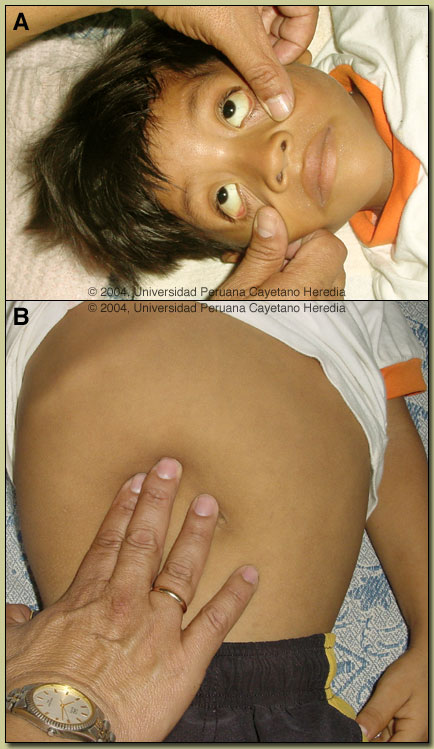
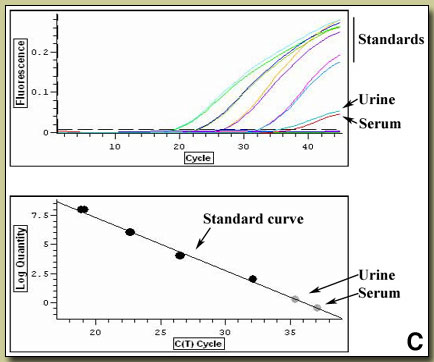
Leptospirosis needs to be considered in the differential diagnosis of any undifferentiated tropical fever. It has protean clinical manifestations and at differing stages of the illness may be impossible to distinguish clinically from yellow fever, dengue, rickettsial disease, typhoid, malaria, brucellosis, tuberculosis, viral hepatitis. The classically described textbook manifestations of leptospirosis represent only a small portion of a much wider spectrum of disease. Typically, leptospirosis is described as having an initial septicemic phase which may be mild, with fever, myalgia, headache, conjunctival suffusion, and abdominal wall pain. The illness may be self-limited but in some cases, after an apparent recovery, may present a biphasic illness and progress to an immune stage manifest by fever, meningitis, and uveitis. The 2 distinct stages may be obscured and run together in severe disease (Weil?s Disease) manifested by jaundice, hemorrhage, and renal failure. In Iquitos as in many other areas of the tropics, data indicates a seroprevalance of up to 40% of the general population (J. Vinetz, unpublished) with most individuals not having a discernible history of a prolonged febrile illness consistent with typical descriptions of leptospirosis. This is consistent with the feeling that most cases in the tropics are not diagnosed due to the non-specificity of the illness and the unavailablity of diagnostic tests in resource poor environments. Our patient did not have the conjunctival suffusion and the muscle tenderness in calves and lumber areas often mentioned as distinguishing features of leptospirosis. A recent review of large case series of leptospirosis found these to be only variably present (40-100%) [Lancet Infect Dis 2003;3(12):757-71]. Typhoid should have responded to the pre-admission ciprofloxacin, there is no brucellosis in the Iquitos area, and the patient was vaccinated against yellow fever and hepatitis. Clinically, patients with viral hepatitis become afebrile with the onset of jaundice. The clinical course here is too long for dengue. Jaundice and bilirubinemia out of proportion with hepatocellular damage is the usual finding in leptospirosis. This is manifest as significant jaundice in the face of an SGOT and SGPT that is no more than 3-4X normal with an alkaline phosphatase that may be as high as 10X normal. The mechanism of the cholestasis in leptospirosis is not entirely clear. The WBC is usually normal to low with no left shift and thrombocytopenia may be present but is not a usual finding. In severe or prolonged disease, renal damage will occur and the sediment is usually active. Isolation of Leptospira in culture is difficult and insensitive. Culture in special media in tubes held at 28-30°C for prolonged periods is necessary. Blood is only positive in the first week of illness after which urine becomes progressively more positive (as in our patient). Cultured leptospires are only visible and confirmed using dark-field microscopy. Diagnosis is most often serological and retrospective (the microscopic agglutination test [MAT], IgM ELISA, or a commercially available dipstick test). PCR-based diagnosis holds great promise due to the rapidity with which results can be available compared to other techniques. Leptospirosis is endemic in almost every country but more so in the tropics. Traditionally an occupational disease and a disease of poverty, it has also emerged as a disease of adventure travelers (hikers, bikers, boaters, swimmers) who have contact with standing or moving water. Leptospires may penetrate conjunctiva, macerated skin, or possibly the oropharynx. Leptospirosis is maintained in the environment by long-term carriage and excretion of the organism from the urinary system of asymptomatic animal carriers. Rodents are most frequently implicated with swine, cattle, and dogs next most frequent, but the full spectrum of mammals forming the reservoir is unclear. Classification of leptospires is complex and obscure to most clinicians. While most human isolates are L. interrogans, further division into a number of species using DNA relatedness is hampered by a traditional naming system that uses serologically defined antigenic determinants (serovars and serogroups) that may be shared by two or more species. Whether or not to treat leptospirosis remains controversial [Clin Infect Dis 2003;36(12):1514-5]. Insufficient numbers of cases from properly controlled trials a |
 Discussion:
Discussion: