-
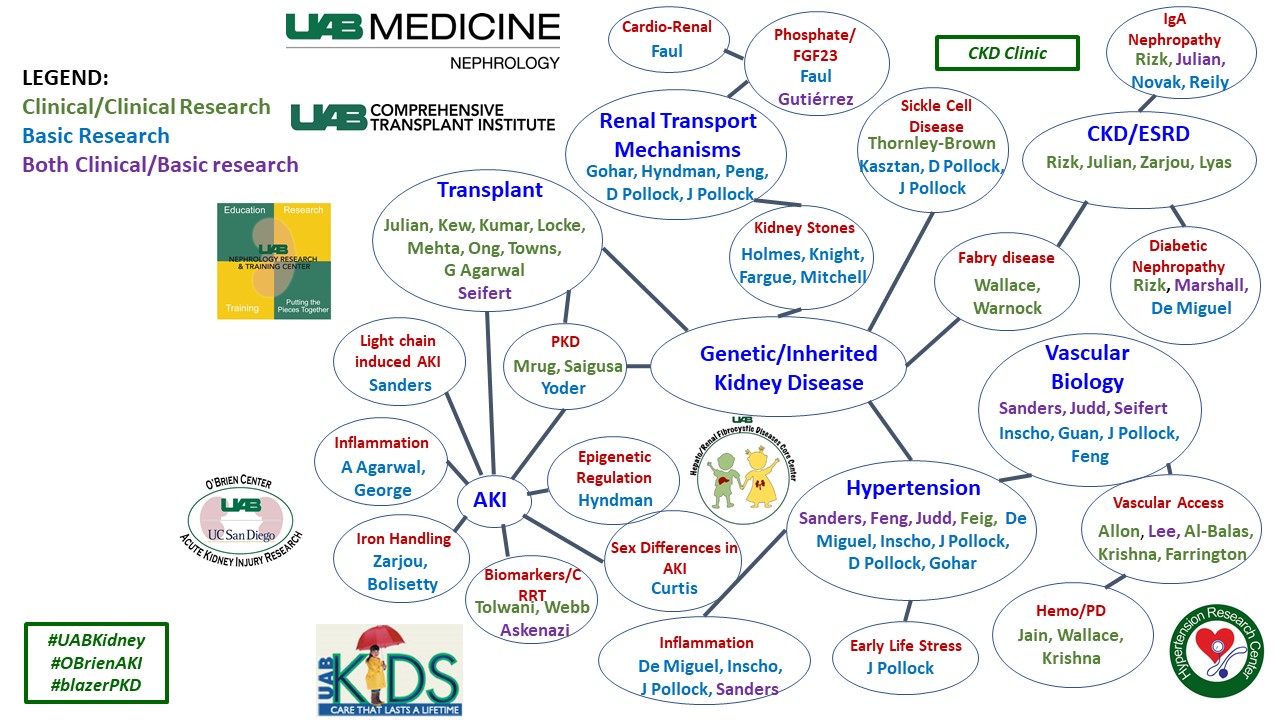
Chronic Kidney Disease and ESRD
Chronic Kidney Disease and ESRD
-
ICU Nephrology and Acute Kidney Injury
ICU Nephrology and Acute Kidney Injury
-
Transplantation
Transplantation
-
Epidemiology and Outcomes
Epidemiology and Outcomes
-
Inherited Kidney Disease
Inherited Kidney Disease
-
Sickle Cell Disease
Sickle Cell Disease
-
Vascular Access
Vascular Access
-
Hypertension
Hypertension
-
Home Hemodialysis and Peritoneal Dialysis
Home Hemodialysis and Peritoneal Dialysis
-
Telemedicine
Telemedicine