 |
Gorgas Case 2026-4 |
 |
|
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by the 2026 Gorgas Course participants.
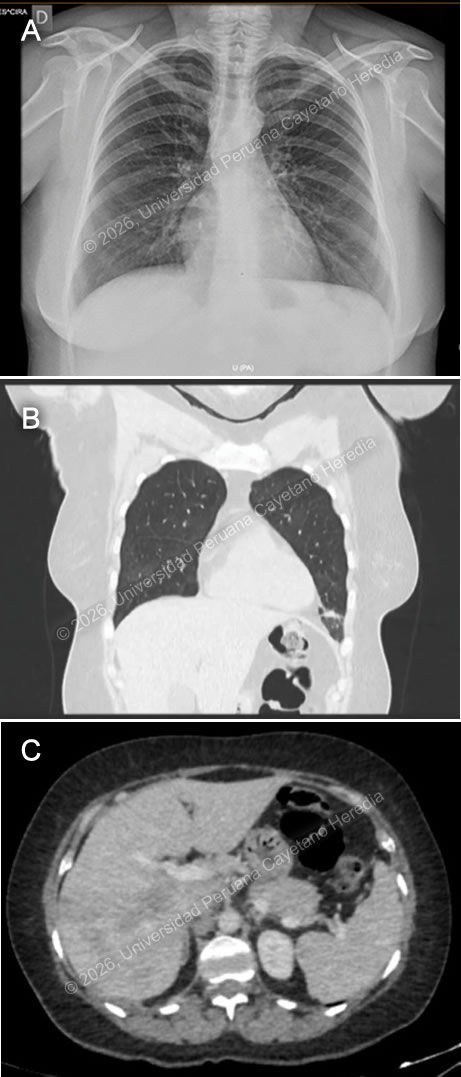 History: A 53-year-old woman presented with a 20-day history of progressively worsening dyspnea and cough. Her symptoms began with mild shortness of breath and a productive cough, accompanied by subjective fever, malaise, and night sweats. Over the following days, her dyspnea intensified, and the cough became blood tinged. On the day of admission, she developed dyspnea at rest, prompting her to seek care in the Emergency Department, where she was subsequently admitted. She reported intermittent abdominal pain over the past two years, unintentional weight loss of five kilograms in the last month, and pruritus of the hands and feet. Epidemiology: The patient is a native of Ancash (Congos–Ocras) and currently resides in Ocros. He works as an agricultural worker and livestock breeder, with frequent direct exposure to domestic animals, including cattle, donkeys, horses, poultry, pigs, sheep, and guinea pigs. He reports consuming home-produced dairy products and watercress since childhood. Physical examination: HR: 82 x’, RR: 22 x’, Temp: 36.6°C, BP: 100/70, SatO2: 92% on room air. On admission, the patient was afebrile, hemodynamically stable, and had low oxygen saturation on room air. Skin examination revealed mild pallor. Respiratory examination showed tachypnea and bilateral wheezing. The remainder of the physical examination was unremarkable. Laboratory: Initial laboratory tests showed a normal hemoglobin of 14.7 and leukocytosis of 36,300; with 15.9% neutrophils, 4% lymphocytes, 1% basophils, 1% monocytes, and marked eosinophilia at 74.4%, corresponding to an absolute eosinophil count of 27,007. Platelet count was 281,000. Liver function tests revealed elevated enzymes; AST 78 U/L (normal value 8 to 48 U/L) and ALT 199 U/L (Normal range 7 to 55 U/L). There was also a significant increase in alkaline phosphatase to 609 (normal range 30 to 130 U/L), and GGT of 143 (normal range 5 to 40 U/L). Total bilirubin was 0.8, with direct bilirubin 0.4. Renal function was within normal limits, with urea 40 mg/dL and creatinine 1 mg/dL. Electrolytes were also normal, including sodium 140 mEq/L, potassium 4.94 mEq/L, and chloride 98 mEq/L. Total protein was 9 g/dL, and albumin was 4.4 g/dL. A five-method stool examination was positive for Blastocystis sp. and Entamoeba coli. Imaging: A chest X-ray shows a reticular pattern in both lung fields (Image A), and a Chest CT scan shows ground-glass infiltrates and fibrotic bands in the bases of both lung fields (Image B). An abdominal CT scan that shows hepatomegaly and the presence of hypodense lesions in the liver parenchyma (Image C). UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
 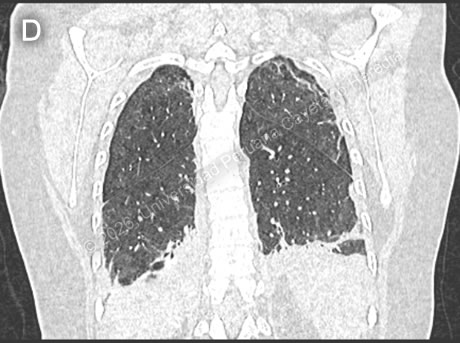 Discussion: A FAS-2 ELISA test was performed, which was positive, confirming the diagnosis of fascioliasis. Fascioliasis is a zoonotic, food-borne parasitic disease caused by the trematodes Fasciola hepatica and F. gigantica, particularly in Africa and Asia. Also known as “liver flukes,” it infects humans as well as a variety of domestic and wild animal hosts. It is acquired by ingesting encysted metacercariae attached to aquatic vegetation, most commonly watercress, in stagnant or slow-moving freshwater environments (1). The highest prevalence is reported in areas where livestock farming is common and raw aquatic plants are frequently consumed, such as South America, Africa, and Asia (2). In the Peruvian highlands, the highest rates are observed in Cajamarca, Cusco, Puno, Junín, and Ancash (3). Clinical presentation depends on the phase of infection. The acute phase is caused by larval migration through the intestine and liver, often involving tissue damage and inflammation; it can present with fever, abdominal pain, and eosinophilia. The chronic phase involves adult flukes in the bile ducts, in some cases for years, causing inflammation and obstruction, which can lead to complications such as cholangitis, cholecystitis, gallstones, liver abscesses, or cirrhosis (4). In acute fascioliasis, eosinophilia occurs in nearly all cases. Our patient exhibited hypereosinophilia and progressively worsening respiratory symptoms consistent with acute eosinophilic pneumonia, as a rare complication previously reported (5), in which eosinophilic infiltration and degranulation lead to lung inflammation and the associated clinical manifestations (6). Other possible laboratory findings include anemia, commonly due to chronic biliary blood loss, and liver enzymes and bilirubin levels are frequently elevated during both larval migration and the chronic stage. For diagnostic confirmation, serology is more sensitive than stool testing, especially in non-endemic regions. Our patient had a positive serology for FAS-2 ELISA and two negative stool Kato-Katz samples. Management of acute eosinophilic pneumonia includes high-dose steroids, which typically improve symptoms within 48 hours (6). Along with addressing the underlying cause. Triclabendazole is the treatment of choice for fascioliasis, with high cure rates and an acceptable safety profile. Although mass treatment has been proposed for control in humans and livestock, inconsistent administration, particularly in resource-limited settings, may promote the development of drug resistance (2). Our patient was treated with triclabendazole 750 mg daily for two days along with corticosteroids. Despite initial worsening of respiratory distress, also evidenced in imaging (Image D), there was marked improvement within 48 hours after starting prednisone at 1 mg/kg per day for one week. Following therapy, the patient showed significant laboratory and rapid clinical recovery, with complete resolution of respiratory failure and eosinophilia. References |