 |
Gorgas Case 2026-6 |
 |
|
The following patient was seen on the inpatient ward of Cayetano Heredia Hospital in Lima by the 2026 Gorgas Course participants.
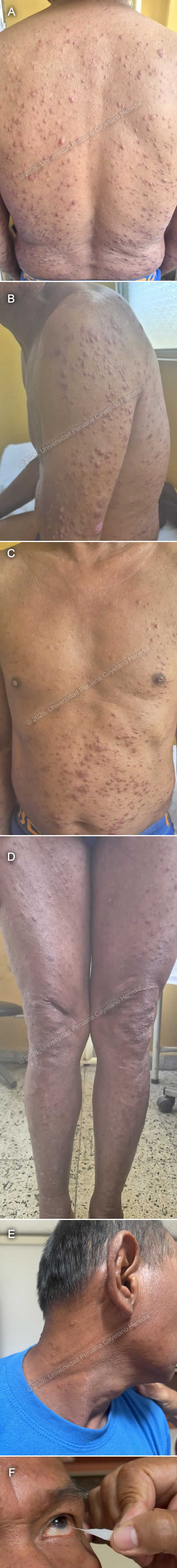 History: A 54-year-old man presents with a six-month history of progressively worsening generalized pruritic skin lesions that initially appeared as hives and papules, affecting the abdomen and upper limbs before spreading to the rest of the body. Three months later, he began experiencing sporadic nosebleeds and increasing numbness in his hands and feet. Due to decreased sensation in his hands, he has developed wounds from unnoticed trauma. In the last month, as his difficulty performing work-related activities worsened, he was advised to seek further medical evaluation. Over the past six months, the patient reports a weight loss of approximately 4 kg without trying. Epidemiology: The patient was born in a rural area of Yurimaguas, in the low jungle of Peru, and has been living in Lima for the past 16 years. He is currently a warehouse worker in Lima. When he was in the jungle, he worked as a farmer for 6 years, cultivating corn, rice, bananas, and cassava, and raising animals including pigs, chickens, and cattle. He reports a past history of cutaneous leishmaniasis at age 12, with complete antimonial treatment. He also mentions having a nasal septum perforation since he was 18, with no clear cause. He denies sick contacts. Physical examination: His vital signs were HR: 78 beats per minute, RR: 18 breaths per minute, BP: 110/70 mmHg, T: 36.7°C, SatO2: 98% on room air. Skin examination revealed multiple very superficial papules and nodules in a dispersed pattern involving his face, abdomen, back, and both upper and lower limbs (Images A, B, C, D). These were associated with nerve thickening (Image E), corneal anesthesia with absence of corneal reflex (Image F), and decreased sensation in the hands and feet. A perforated septum was also observed. The rest of the exam was unremarkable. Laboratory: Initial tests showed hemoglobin at 13.2 g/dL and leukocytes at 5530, with 68.9% neutrophils, 22.2% lymphocytes, 0.2% basophils, 7.6% monocytes, and 1.1% eosinophils. Platelet count was 437000. Liver function tests revealed AST at 17 U/L (normal range 8 to 48 U/L) and ALT at 10 U/L (Normal range 7 to 55 U/L). Total bilirubin was 0.75, with direct bilirubin at 0.3. Renal function was within normal limits, with urea at 21 mg/dL and creatinine at 1.02 mg/dL. Glucose was 68 mg/dL. UPCH Case Editors: Carlos Seas, Course Director / Paola Nakazaki, Associate Coordinator |
|
  Discussion: A skin punch biopsy from the lesions on the back was performed, revealing a moderate inflammatory infiltrate in the dermis with a nodular and linear pattern. It consisted of vacuolated histiocytes containing multiple bacillary structures. Fite–Faraco staining (Image G) is positive (3+), confirming the diagnosis of lepromatous leprosy. Leprosy is a chronic granulomatous infection caused by Mycobacterium leprae. It remains a neglected tropical disease present in over 120 countries, with approximately 200,000 new cases each year. Although it was declared eliminated as a global public health problem by 2000, new cases still appear in countries like Brazil, India, and Indonesia, while many nations now report few or no cases (1). In Peru, leprosy is endemic in Amazon regions, especially in the departments of Ucayali, Loreto, Amazonas, Huánuco, and San Martín (2). Twenty six new cases were reported in 2023, all of them from the departments of Loreto and Ucayali (3). Transmission is unclear, though it is thought to occur via respiratory droplets from the nose and mouth of untreated individuals during prolonged close contact (1,4). The incubation period is variable, from months to years, and it commonly affects peripheral nerves, skin, and mucosa (4,5). And if left untreated, it can lead to severe disability, mostly secondary to impairment of nerve function or visible deformity. Clinical manifestations vary among patients due to differences in individual immune responses, and they are classified into Tuberculoid (TT), Lepromatous (LL), Borderline Lepromatous (BL), Mid Borderline (BB), and Borderline Tuberculoid (BT) (6). Lepromatous leprosy is linked to a Th2-dominant immune response to the pathogen and typically presents with widespread, diffuse infiltration of the skin and very superficial papules and nodules (lepromas) containing numerous organisms, along with nerve involvement (5). Diagnosis is made through clinical history and examination, with slit-skin smear microscopy or biopsy with histopathology as needed. Diagnosis requires at least one of the following criteria: sensory loss in skin lesions, thickened peripheral nerves, or detection of acid-fast bacilli on slit-skin smears (7). The absence of reliable point-of-care tests often contributes to delayed diagnosis and increased risk of nerve damage and disability (1,5). WHO guidelines for treatment recommend a three-drug regimen with rifampicin, dapsone, and clofazimine with variable duration depending on the clinical category of presentation, with a recommended 12-month regimen for multibacillary leprosy (1). Monthly RMM is the standard in the US and in countries where clofazimine cannot be obtained (8). Our patient has multibacillary lepromatous leprosy and was started on rifampicin, dapsone, and clofazimine. He is currently undergoing his second month of treatment with no adverse events (Images H, I). Complications can persist even after bacteriological cure and include immune reactions, nerve damage, ocular sequelae, and disabilities. Management is not limited to medication, it also needs long-term support to address the physical, psychological, and social impacts of the disease (9). We would like to thank Dr. David Freedman for his contributions. References |