 |
Gorgas Case 2020-03 |
 |
|
The following patient was seen as an outpatient in the Infectious Disease Department of Hospital Cayetano Heredia during the first and second week of the Gorgas Diploma Course.
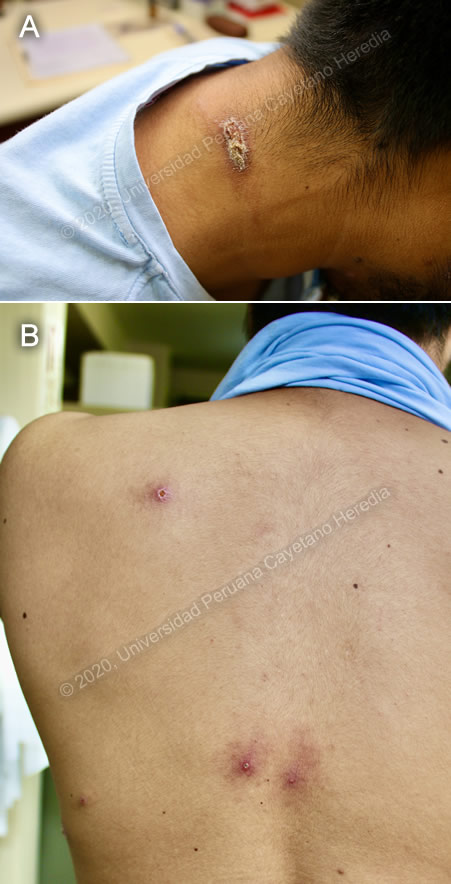 History: A 27-year-old male patient complains of 3 months of ulcerative lesions on the left forearm and nuchal region. The lesions began as small, pruritic, erythematous papules, both of which ulcerated over about two weeks. He was seen by a dermatologist and treated with oral cefradine and topical fusidic acid for 7 days, followed by oral ciprofloxacin and clindamycin for an additional 7 days, without improvement. He noticed the appearance of multiple new painless ulcers on his shoulders and back and presented to our institution. Epidemiology: He works as a botanist. Two months before symptom onset, he traveled to La Convencion, Cusco (tropical jungle) to work on an environmental impact study, where he camped outdoors, bathed in rivers and got many arthropod bites. The next month, he traveled for work to Loreto (tropical jungle) for a month under similar conditions. He denies any previous illnesses or contact with TB patients. No other colleagues had presented with similar symptoms. Physical Examination: BP: 120/80 mmHg; RR: 16; HR: 65; afebrile. 18 different skin ulcers (as pictured in Images A and B) on the face, neck, torso, arms, legs, and genitals. The skin lesions pictured are painless ulcers with elevated borders, with the center covered in white-yellow fibrinous material. The rest of the physical exam was unremarkable. Laboratory: Hb:14.8 g/dL; WBC 8.85 (bands: 0%, neutrophils: 53%, eosinophils: 2%, basophils: 0%, monocytes: 6%, lymphocytes: 39%); Platelets: 302 000. Urea: 44 mg/dL, Creatinine: 1.04 mg/dL, AST: 22 U/L, ALT 20 U/L, HIV: non-reactive. |
|
Diagnosis: Disseminated cutaneous leishmaniasis 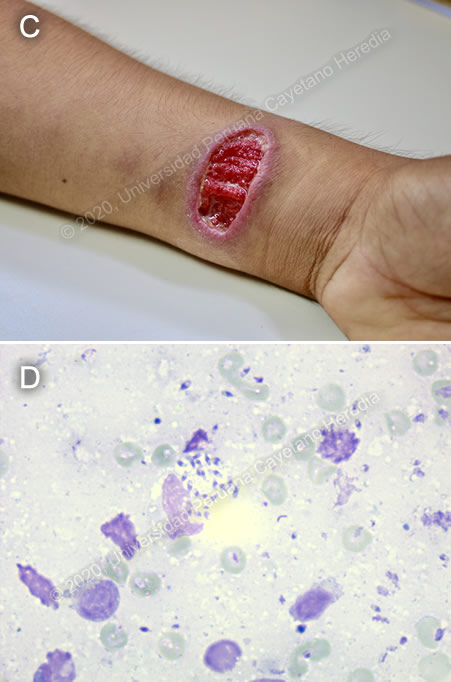
 Discussion: Image C is typical of new world leishmaniasis. Microscopy of a skin scrapings with Giemsa staining revealed numerous Leishmania sp. amastigotes (Image D). A leishmanin skin test (LST) was positive. PCR for Leishmania sp. speciation was not available. About 74% of Peru is endemic for leishmaniasis, which is transmitted by the bite of the female sandfly: Phlebothomus sp. for Old World Leishmaniasis and Lutzomyia sp. for New World Leishmaniasis. Two forms are typically described. The Andean form, commonly known as “uta”, occurs on the western slopes of the Andes at altitudes between 900 and 3000 meters above sea level and are usually caused by L. peruviana. The Sylvatic form, or “espundia”, occurs in the tropical jungle and is usually caused by L. braziliensis. Other species that have been identified in the Peruvian territory include L. guyanensis, L. lainsoni, L. amazonensis, and L. shawi (https://www.ncbi.nlm.nih.gov/pubmed/9715953, https://www.ncbi.nlm.nih.gov/pubmed/31220120). In 2018, the Ministry of Health reported 6022 cases of leishmaniasis, of which 92.5% was cutaneous and 7.5% was mucocutaneous (https://www.dge.gob.pe/portal/docs/vigilancia/sala/2019/SE07/leishmaniosis.pdf) The clinical manifestations of leishmaniasis are diverse and are influenced by the infecting species. Localized cutaneous leishmaniasis presents as one or a few characteristic lesions, as described above, with a positive LST. In Peru, it is typically caused by L. peruviana and L. amazonensis. Mucocutaneous leishmaniasis, as the name states, involves mucosal membranes and may be disfiguring. Most cases with mucosal involvement in Peru are related to L. braziliensis, which typically start as cutaneous lesions that later progress, often years after resolution of the primary lesions, destroying mucosal tissue. Diffuse cutaneous leishmaniasis, a rare form of the disease, presents as multiple nodular lesions that do not form ulcers, and is characteristically associated with anergy, with a negative LST (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5486117/). Our patient had a clinical form of leishmaniasis known as disseminated leishmaniasis which was first described in patients from the northeastern region of Brazil (https://www.ncbi.nlm.nih.gov/pubmed/12447770?dopt=Abstract). Patients present with an initial ulcerated lesion, that erupts into multiple pleomorphic lesions within 2-6 weeks, with no further exposure to the parasite from subsequent mosquito bites. Patients may present non-specific systemic symptoms such as fever, asthenia, or nausea. The lesions may be acneiform, papular, nodular, or ulcerated, ranging from dozens to hundreds. Lesions are found in two or more non-contiguous areas of the body, and commonly involve mucosal membranes (in up to 53% of cases) (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6360961/). This clinical form seems to stem from a complex interaction between host and parasite factors. Peripheral blood lymphocytes from patients with disseminated disease seem to produce lower levels of Th1 than in patients with localized disease, but immune function at the lesion site is preserved. On the other hand, L. braziliensis isolated from patients with disseminated leishmaniasis have a higher production of TNF and INFγ than isolates of the same species from patients with localized disease (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5486117/). In patients presenting with disseminated cutaneous lesions, the differential includes other infections such as cutaneous histoplasmosis, paracoccidioidomycosis, myiasis, and tuberculosis, which are all endemic in Peru. In other parts of the world, one might also consider talaromycosis and eschars of scrub typhus. However, for all of the conditions mentioned, one would have to look for coexisting immunosuppressive conditions (HIV infection, use of biological therapy), which was not the case for our patient. The diagnosis of disseminated leishmaniasis relies on clinical characteristics, and laboratory confirmation is based on the identification of the parasite by direct observation, culture, PCR or histopathology. Lesions typically have a large number of amastigotes and are easily identifiable in skin scrapings. Skin scrapings are preferred over punch biopsies because they are rapid, non-invasive and require less expertise. In this type of sample, amastigotes might be seen outside macrophages (as seen in Image D), unlike in skin biopsies. LST is positive in up to 83% of cases (https://www.ncbi.nlm.nih.gov/pubmed/21723576/). Also called Montenegro test, LST is an immunologic skin test that measures delayed-type hypersensitivity (DHT) to Leishmania antigen; it is not available in most countries. Intradermal injection of killed promastigotes will induce a DHT reaction in individuals with current or prior exposure to Leishmania. Injection site is examined 48 hours later and an induration of ≥ 5 mm is considered positive. Disseminated leishmaniasis is a challenge to treat, and has low cure rates (23%). Pentavalent antimonials are the first-choice therapy, but patients typically require more than one course of treatment or switching to amphotericin B (http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0365-05962019000100002). Liposomal amphotericin B has shown promising results for treatment of this condition, with cure rates around 80-90% and more tolerable side effects, but is expensive and difficult to obtain in Peru. Our patient is currently receiving a course of amphotericin B deoxycholate for a total cumulative dose of 25mg/kg, showing moderate improvement of skin lesions. Due to its toxic effects, the patient is being administered the treatment every other day. After course completion, the patient will be seen at the outpatient clinic in order to evaluate the need for further treatment courses. |