 |
Gorgas Case 2020-04 |
 |
|
The following patient was seen in the surgical intensive care unit at Hospital Cayetano Heredia during the Gorgas Diploma Course.
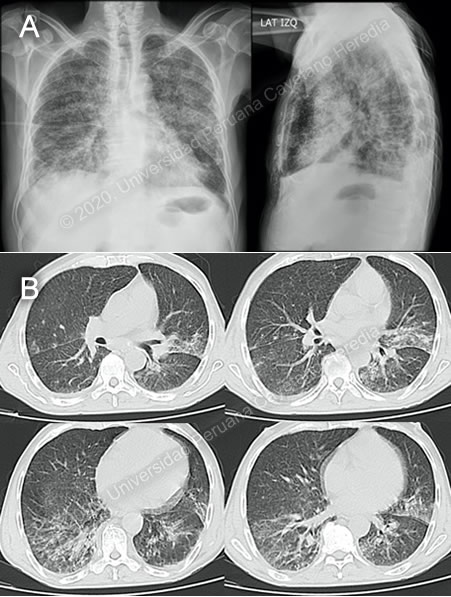 History: A 60-year-old male patient presented to the hospital complaining of 10 weeks of fever, night sweats and weight loss. 8 weeks before admission, he presented to a local physician with mild diffuse abdominal pain, and watery diarrhea, and is treated with ciprofloxacin for 7 days, with remission of the diarrhea. Over the next 2 weeks, he develops anorexia and asthenia, accompanied by worsening abdominal pain. He is treated with ivermectin, but his symptoms show no improvement. A month prior to admission, he starts noticing dyspnea, which steadily progresses and by the day of admission to the Tropical Medicine ward is present at rest. Epidemiology: Born and lives in Moyobamba, San Martin (tropical jungle of Peru). Worked as a farmer until 2005 but is currently unemployed. Raises chickens around his house and also has dogs. Past medical history includes cryptococcal meningitis in 2008. The patient denies other past illnesses and surgeries. No known TB contacts. Physical Examination: BP: 90/70 mmHg; RR: 24; HR: 116; T: 38°C. Presented with marked pallor; no rash or petechiae. No mucosal lesions. Palpable bilateral inguinal lymphadenopathy, about 3cm in diameter. Pulmonary exam revealed bilateral diffuse crackles. Cardiovascular exam was unremarkable. Abdomen was not distended but slightly tender to palpation, liver span ±15cm, no splenomegaly. Patient was alert, oriented, with no focal deficits, and no meningeal signs. Imaging Studies: CXR revealed bilateral interstitial infiltrates (Image A). Thoracic CT showed interstitial infiltrates in both inferior lobes and the left superior lobe, with bilateral bronchiectasis and mediastinal lymphadenopathy(Image B). Abdominal CT revealed discrete hepatomegaly. Laboratory: Hb 8.9 g/dL; Hct 28%; WBC 4.64 (bands 0%, neutrophils 71.1%, eosinophils 1.3%, basophils 0.2%, monocytes 9.3%, lymphocytes 16.8%); Platelets 163 000. Gluc 103 mg/dL, Urea 37 mg/dL, Creat 0.7 mg/dL, Na 133 mEq/L, K 3.57 mEq/L, Cl 99 mEq/L, Total bilirubin 0.3 mg/dL, Indirect bilirubin 0.2 mg/dL, Alk phos 169, AST 108, ALT 50, GGT 107, LDH 291 (N=???), CRP 197.7, HIV and HTLV non-reactive. Blood cultures x2: negative. AFB, GeneXpert MTB/RIF, culture in BAL for bacteria and mycobacteria: negative. |
|
Diagnosis: Progressive disseminated histoplasmosis 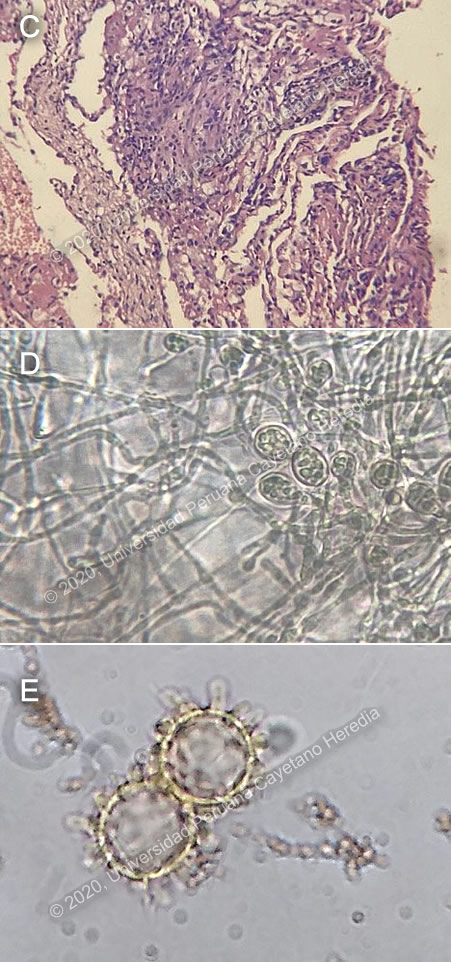
 Discussion: A transbronchial lung biopsy revealed chronic granulomatous infiltration with some giant multinucleated cells; Ziehl-Neelsen and Grocott stains were negative for AFB and fungi (Image C). Bone marrow culture was positive for Histoplasma capsulatum, showing multiple macroconidia stemming at right angles from the parent hyphae (Image D). Bone marrow biopsy showed a granulomatous reaction with no evidence of malignancy. Image E shows the typical 7-10 μm H. capsulatum tuberculated macroconidia, after 30 days of growth. Urine histoplasma antigen was positive as well. H. capsulatum is a dimorphic endemic fungus of worldwide distribution, most commonly reported in the Americas. It has been estimated to have an incidence of less than 0.1 per 100 000 inhabitants in Peru (https://www.ncbi.nlm.nih.gov/pubmed/28188491). H. capsulatum has been isolated from pigeon droppings in Lima, the country’s capital, (https://repositorio.cientifica.edu.pe/handle/UCS/436), and from the soil in caves in Tingo María, in the tropical Peruvian jungle, which has been linked to histoplasmosis outbreaks in tourists (http://www.scielo.org.pe/scielo.php?pid=S1726-46341955000100003&script=sci_arttext). It is presumed to be endemic in all of the Peruvian jungle. Infection occurs through inhalation, and has been linked to occupational and recreational activities that disrupt soil or bird and bat droppings, such as, in the case of our patient, agriculture. Although exposure to H. capsulatum is very common in endemic areas, as is evidenced by high cutaneous histoplasmin positivity rates, overt clinical manifestations occur in less than 1% of infected people. The most common manifestation is acute pulmonary histoplasmosis, a self-limited illness that presents with fever, malaise, headaches, and weakness. Nonetheless, patients with immunosuppression may present with disseminated disease. Upon infection, most patients have asymptomatic hematogenous dissemination through parasitized macrophages and, although, immunocompetent hosts are able to control the infection through T-lymphocyte mediated macrophage activation, they may still retain foci of H. capsulatum in various organs which can reactivate if there is altered immunity. Thus, even patients who have not been in endemic areas for many years may present the disease. Disseminated histoplasmosis is mostly seen in immunocompromised patients (HIV, patients on corticosteroids or biological therapy, extremes of ages), however, it has also been reported in patients with no identifiable risk factors. In the setting of a patient with disseminated histoplasmosis with no clear cause of immunosuppression, experts recommend further study of immune status, i.e lymphocyte and immunoglobulin studies. The disease typically presents with malaise, anorexia and weight loss. Physical exam may reveal hepatosplenomegaly, lymphadenopathy, pallor, and mucocutaneous ulcers, nodules or papules. Laboratory studies may show non-specific alterations such as elevated alkaline phosphatase, pancytopenia, and elevated inflammatory markers. Gastrointestinal compromise is frequent, presenting with abdominal pain and diarrhea, though most cases are asymptomatic. Suprarenal glands are often affected, causing fever, malaise, orthostatic hypotension, nausea, vomiting, and electrolyte abnormalities. Mucosal involvement is frequent, with ulcerations in tongue, gingival and buccal mucosa, lips, pharynx, and larynx. Every organ system can be affected, but many are rarely symptomatic. The diagnosis of disseminated histoplasmosis is made by microscopic evidence or isolation of H. capsulatum from extrapulmonary sites. Cultures may be drawn from the blood, lungs, bone marrow, or any other suspected site, and are the gold standard for diagnosis, but take a long time to grow. Fully formed macroconidia, as seen in the images for this case, are only seen after 30 days. Cultures typically have a higher yield in patients with disseminated disease. Biopsies from affected organs often reveal typical 2-4 μm intracellular yeasts with a narrow base budding, best seen with PAS or methenamine silver stains. Rapid diagnosis can be achieved by means of antigen detection by enzyme immunoassay in urine, serum, cerebrospinal fluid or bronchoalveolar lavage. Urine antigen testing is positive in about 75% of immunocompetent patients and 95% of immunocompromised patients with disseminated histoplasmosis, but may be falsely positive in the setting of infection with other endemic mycoses (https://www.ncbi.nlm.nih.gov/pubmed/21810734). Serologic testing is most useful for subacute and chronic forms of infection, as antibodies become detectable after 4-8 weeks of infection. However, positive serology only indicates exposure to the fungus at some point and is of little use in endemic areas, where most people have been exposed. The differential diagnosis for a patient presenting with a long history of fever, lung and abdominal involvement must include tuberculosis, especially in endemic areas such as Peru. The clinical presentation of TB may be indistinguishable from that of disseminated histoplasmosis. Molecular testing for tuberculosis has a relatively high sensitivity and specificity —GeneXpert MTB/RIF in bronchoalveolar lavage has a sensitivity of 80% and a specificity of 98% (https://www.ncbi.nlm.nih.gov/pubmed/29964143), which helps to narrow down the diagnosis. Endemic mycoses such as cryptococcosis or talaromycosis (in the right endemic setting) should also be considered in the differential diagnosis; as for non-infectious differential, also include malignancy and other inflammatory diseases such as sarcoidosis. Treatment is warranted in all cases of disseminated histoplasmosis, as the natural history of this form of infection is almost invariably fatal. Patients with severe disease should be initially treated with a lipid formulation of amphotericin B, which quickly eradicated fungemia and has more easily titratable serum levels than itraconazole. If there is a good response, after one or two weeks and if there is no fever and good oral tolerance, patients can be switched to itraconazole. Fluconazole, posaconazole and voriconazole may be used if itraconazole is not tolerated, but these agents have not been as well studied. Treatment should be continued for at least a year, to reduce the risk of relapse (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1797635/). Our patient was empirically started on amphotericin B and the standard anti-tuberculosis regimen. When cultures came back positive for H. capsulatum, tuberculosis drugs were suspended and amphotericin B continued. Further testing revealed a CD4 count of 37/uL (18%) and CD8 count of 145/uL (72%), with a CD4/CD8 of 0.26. Unfortunately, he did not respond well to treatment and succumbed to the infection. |