 |
Gorgas Case 2021-01 |
 |
|
The Gorgas Courses in Clinical Tropical Medicine are usually given at the Tropical Medicine Institute at Cayetano Heredia University in Lima, Peru. This year, due to the pandemic, we have started the 26th edition of the Gorgas Course in Clinical Tropical Medicine in a mixed educational model involving a virtual format, which will be followed by a live session at the Tropical Medicine Institute. New cases will be published every two weeks for the next seventeen weeks. Each case includes a brief history and digital images pertinent to the case. A link to the actual diagnosis and a brief discussion follows.  History: A 51-year-old female patient presented with a 6-month history of non-productive cough. Two weeks prior to admission the patient noted worsening of her cough, as well as subjective fevers, shortness of breath on exertion, and back pain. The patient continued to deteriorate to the point of having shortness of breath on rest for which she decided to go to the Emergency Department at Cayetano Heredia Hospital for further evaluation. Epidemiology: Born in Iquitos, a city in the Amazon jungle; has lived in Lima for the past 2 years. Works as a homemaker. Her father had tuberculosis 10 years ago. Physical Examination: BP:90/60, HR 92, RR 32, T 38.9°C. SatO2: 89% (FiO2 100% - reservoir mask). Skin and mucosae were pale. Patient was tachypneic; lung examination revealed chest retractions, diminished breath sounds in both bases and diffuse crackles. Cardiac and abdominal examinations were unremarkable. The patient was alert, oriented, without significant neurological deficits. Imaging Studies: Chest x-ray showed bilateral alveolar infiltrates, air bronchograms, and a lesion in upper left lobe (Image A). Chest CT showed bilateral diffuse alveolar infiltrates, with air bronchograms (Image B). Laboratory: Hb: 11.2 g/dL; Hct. 33%; WBC 11 880 (neutrophils: 93%, eosinophils: 0.1%, lymphocytes: 0.4%); Platelets: 284 000. ABG: pH 7.2, PO2 81.7, PCO2 65.4, Pa/Fi 148, HCO3 25.9, Lactate 1.8. INR 1.19, PT 15.6, PTT 49.5. Gluc: 115 mg/dL, Urea: 74 mg/dL, Creat: 1.6 mg/dL, AST 85, ALT N/A, GGT 169, Alk Phos 281, LDH 565, albumin 1.8. HIV, HBsAg, HTLV-1/2 were non-reactive. VDRL negative. Antibody test for COVID-19 was IgG positive but PCR was unavailable. A sputum sample was sent to the laboratory.
|
|
Diagnosis: Bilateral pulmonary tuberculosis and Strongyloides hyperinfection. 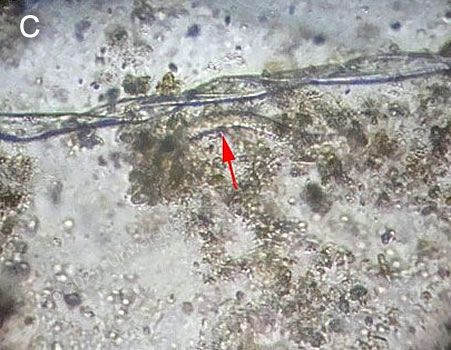
 Discussion: Shortly after admission, the patient deteriorated and required intubation and mechanical ventilation. She was started on empiric treatment with dexamethasone for severe COVID-19. AFB in sputum was positive 2+, and GeneXpert MTB/RIF detected M. tuberculosis without rifampicin resistance. Two weeks after admission, because she was not improving after initiation of 4-drug TB therapy, a bronchial aspirate was sent to the lab, and filariform larvae of Strongyloides sp. were found (Image C), and a diagnosis of Strongyloides hyperinfection was made. Not all patients presenting with respiratory distress amid the pandemic will have COVID-19 and it cannot be ruled in or out using a rapid antibody test. Rapid antibody tests should not be used for diagnostic purposes, as they are not useful to differentiate acute disease from past infection, or even from infection with other coronaviruses. In the presented case, it is likely that the patient had positive IgG due to a past infection. Bilateral involvement is an unusual presentation of pulmonary tuberculosis in immunocompetent individuals. One observational study found that around 35% of patients with pulmonary tuberculosis had bilateral infiltrates (1)." All investigations for risk factors for immunosuppression (HIV, HTLV, neoplasms) besides use of corticosteroids have so far been negative for the present patient. Strongyloidiasis, on the other hand, is an endemic infection caused by the nematode Strongyloides stercoralis in tropical and subtropical regions. It is hard to estimate its prevalence because the larvae are not easily seen by direct stool examination without concentration techniques; however, some studies estimate a worldwide prevalence of about 8.1% (2). A study conducted in a rural community in the Peruvian Amazon found that 8.7% of the analyzed stool samples had S. stercoralis larvae, and 72% of the analyzed serum samples had a positive ELISA (3). S. stercoralis has both free-living and parasitic stages. Adult female worms live in the human small intestine, laying eggs that hatch into rhabditiform larvae, which are shed in the stool. They then grow into either infective filariform larvae or free-living adults. Filariform larvae can infect humans transcutaneously. They enter the bloodstream, are carried to the lungs and are then swallowed to enter the digestive tract. However, some rhabditiform larvae may mature into filariform larvae prior to being shed and may re-infect the host by invading either the intestinal wall or the perineum. This is denominated an autoinfective cycle. The use of corticosteroids at any dose is a well-known risk factor for Strongyloides hyperinfection, though the dose and duration of treatment that confer increased risk are unclear. A study in Thailand suggested that there was more risk with a duration of therapy of less than 50 days, with a median daily prednisolone-equivalent dosage of 40mg, which would be equivalent to 6mg dexamethasone (the recommended daily dose for severe COVID-19) (4). This effect might be mediated by inhibition of eosinophil and lymphocyte activation, or by increasing the fertility of adult female worms. Hyperinfection may occur as early as 20 days after starting corticosteroid therapy, or as late as years later (5). Cases of Strongyloides hyperinfection secondary to corticosteroids given for COVID-19 have been reported in the literature (6,7). Given that fatality rates for this syndrome can be as high as 70-100%, patients at risk of exposure to Strongyloides should be screened or pre-emptively treated with ivermectin (8). Most disseminated or hyperinfected Strongyloides cases in our institution are HTLV-1 associated. This patient deviates from this pattern, and we are still looking for more evidence of immune suppression. The patient was started on oral ivermectin and standard treatment for tuberculosis. She improved and is now being weaned off a tracheostomy. References: |