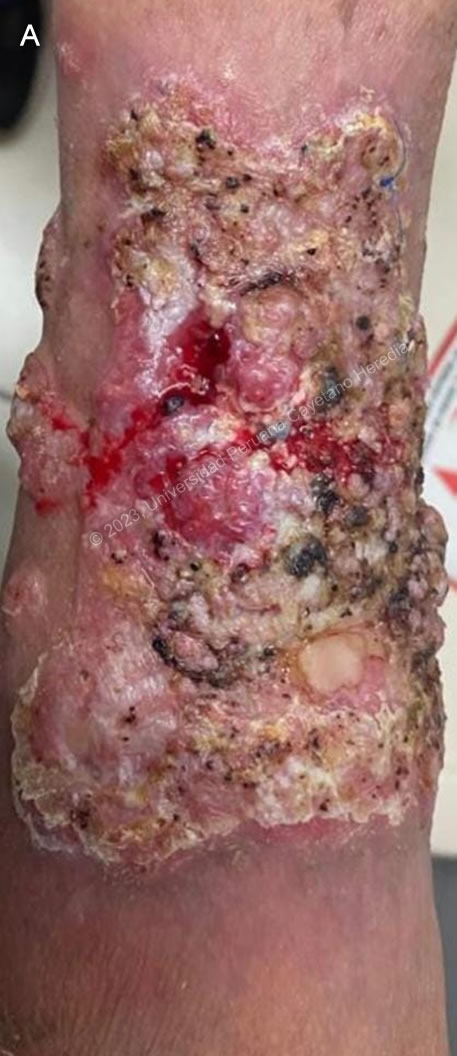
|
Diagnosis: Chromoblastomycosis
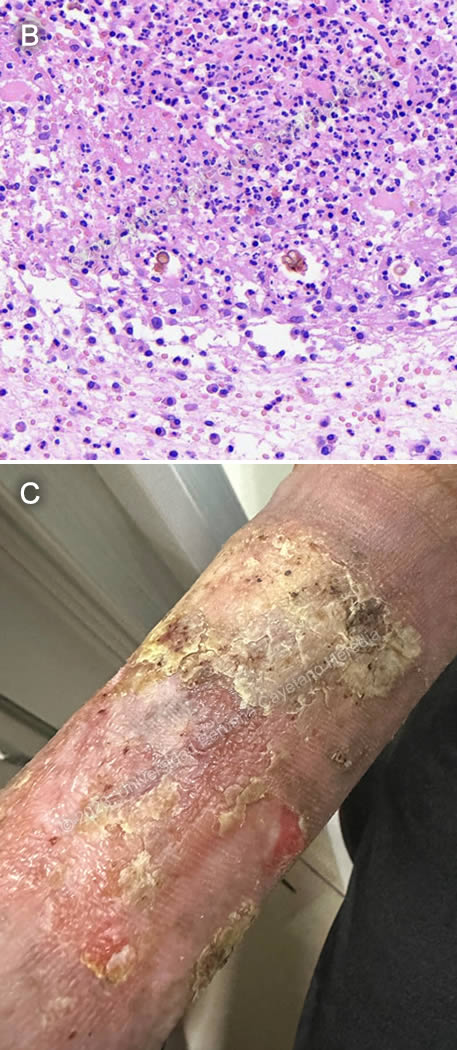
Discussion: Microscopic examination of a skin biopsy revealed chronic granulomatous inflammation with muriform bodies (Image B), which are characteristic of chromoblastomycosis. Fungal cultures for speciation are pending.
Chromoblastomycosis is a chronic subcutaneous mycosis caused by inoculation of spores from dematiaceous fungi. It is prevalent in Southern Africa and parts of Latin America, including Mexico, Brazil and Venezuela. However, it has also been reported in countries with temperate climates. In Peru, sporadic cases have been reported in Ucayali and Cusco1.
It is typically caused by two genera of dematiaceous (also known as melanized or phaeoid) fungi: Cladophialophora and Fonsecaea, both found in tropical rainforests. About 90% of isolated species are identified as Fonsecaea pedrosoi2. They are typically isolated from soil and plants; thus, farm workers, gardeners, and people working with timber (such as in the case of our patient) are at higher risk of infection.
Chromoblastomycosis presents with an initial papule with centrifugal growth that may evolve into different clinical forms. The verrucous-type lesion, as seen in our patient, is the most prevalent. These types of lesions are hyperkeratotic and warty in appearance, have a high fungal load, and ulcerate easily. Another type of lesion is the nodular type, with fibrotic, red-violet nodules with a smooth or hyperkeratotic surface. Plaque-type lesions can also be red or violet, and are usually well circumscribed although irregular in shape, with sharp, elevated edges. The tumoral-type lesions can be single or multiple and coalescent, and can be either smooth or scaly. Cicatricial-type lesions can have an annular, serpiginous, or irregular configuration, and usually grow centrifugally. Metastatic lesions may appear by contiguity, hematogenous or lymphatic dissemination. Lesions are more frequently found in lower limbs, but can appear anywhere in the body. All of the forms typically have a “cayenne pepper” appearance, as seen in Image A. The black dots represent transdermal elimination of fungal debris. Patients are usually oligosymptomatic, presenting only mild pruritus and sometimes local pain, and take a long time to seek medical attention2. Unlike other fungal infections, chromoblastomycosis does not typically affect deeper tissues. In the past 50 years, only two cases of deep tissue invasion in chromoblastomycosis have been reported in Latin America and the Caribbean3.
Diagnosis is made by direct microscopy using KOH of skin scrapings taken from areas of the lesion with abundant black dots, which reveals muriform or sclerotic bodies - pathognomonic for chromoblastomycosis. Skin biopsy (which should include black dots as well) also reveals muriform bodies, along with signs of pyogranulomatous inflammation. Muriform bodies are usually seen as an aggregation of 2-4 round brownish thick-walled 4-12 μm structures with transverse and longitudinal septation. Occasionally, dematiaceous hyphae may also be seen. Fungal culture must be performed in order to identify species; oftentimes molecular biology techniques such as PCR are needed to complete work-up.
The differential diagnosis for a verrucous plaque such as the one seen in our patient includes cutaneous tuberculosis, non-TB mycobacterial infections, sporotrichosis, leishmaniasis, and leprosy. Mycetoma can also present with a verrucous hyperkeratotic plaque, but the lesions develop characteristic sinus tracts with grains, which are not seen in chromoblastomycosis. Phaeohyphomycosis is also caused by dematiaceous fungi but presents as invasive disease with an aggressive course in immunocompromised patients. Non-infectious diagnoses such as sarcoidosis or cutaneous malignancies must also be ruled out.
Treatment is difficult, with low cure and high relapse rates, and there is no high-grade evidence to support it. Therapy choice depends on disease severity. Mild disease, defined as a solitary plaque or nodule less than 5cm in diameter, should be treated with surgical excision, which can be curative. Cryotherapy and thermotherapy may also be considered. However, since most patients seek medical care late in the course of the disease, this is not often an option. Moderate disease includes single or multiple lesions with nodular, verrucous or plaque morphology, less than 15cm in diameter, and involving up to two adjacent skin areas. Severe disease occurs when there is extensive involvement. For moderate to severe disease, extended regimens of oral antifungals are recommended. The agents with greatest proven efficacy are itraconazole and terbinafine, given for 6-12 months at high doses, with similar cure rates of 15-80%4. One study conducted among patients in Brazil showed that terbinafine was the most active antifungal agent (with the lowest MIC), followed by itraconazole and voriconazole5. In cases of resistance to standard antifungals, posaconazole has shown good clinical success rates6. Combination therapy may also be considered in cases of resistance, or in severe or invasive infections, though few studies have assessed antifungal activity against melanized fungi. One study found a synergic interaction between terbinafine and itraconazole against Cladophialophora carrionii, but neither synergy nor antagonism for Phialophora verrucosa nor Fonsecaea pedrosoi. There is also clinical data that suggests that the combination of itraconazole and 5-flucytosine may be effective7.
Complications of chromoblastomycosis include tissue fibrosis that may lead to lymphatic blockage and stasis, bacterial superinfection, or even development of malignancy. Some cases of chromoblastomycosis-associated squamous cell carcinoma have been reported8.
Our patient is currently in his fifth month of treatment with oral itraconazole and is also receiving cryotherapy with liquid nitrogen. The lesion is slowly improving (Image C).
References:
1. Ramírez Soto MC, Malaga G. Subcutaneous mycoses in Peru: a systematic review and meta-analysis for the burden of disease. International Journal of Dermatology. 2017;56(10):1037-1045. doi:10.1111/ijd.13665
2. Brito AC de, Bittencourt M de JS. Chromoblastomycosis: an etiological, epidemiological, clinical, diagnostic, and treatment update. An Bras Dermatol. 2018;93:495-506. doi:10.1590/abd1806-4841.20187321
3. Guevara A, Siqueira NP, Nery AF, Cavalcante LR da S, Hagen F, Hahn RC. Chromoblastomycosis in Latin America and the Caribbean: Epidemiology over the past 50 years. Medical Mycology. 2022;60(1):myab062. doi:10.1093/mmy/myab062
4. Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev. 2017;30(1):233-276. doi:10.1128/CMR.00032-16
5. de Andrade TS, de Almeida AMZ, Basano S de A, et al. Chromoblastomycosis in the Amazon region, Brazil, caused by Fonsecaea pedrosoi, Fonsecaea nubica, and Rhinocladiella similis: Clinicopathology, susceptibility, and molecular identification. Med Mycol. 2020;58(2):172-180. doi:10.1093/mmy/myz034
6. Negroni R, Tobón A, Bustamante B, Shikanai-Yasuda MA, Patino H, Restrepo A. Posaconazole treatment of refractory eumycetoma and chromoblastomycosis. Rev Inst Med Trop Sao Paulo. 2005;47(6):339-346. doi:10.1590/s0036-46652005000600006
7. Queiroz-Telles F, de C L Santos DW. Challenges in the Therapy of Chromoblastomycosis. Mycopathologia. 2013;175(5):477-488. doi:10.1007/s11046-013-9648-x
8. Azevedo CMPS, Marques SG, Santos DWCL, et al. Squamous cell carcinoma derived from chronic chromoblastomycosis in Brazil. Clin Infect Dis. 2015;60(10):1500-1504. doi:10.1093/cid/civ104
|