 |
Gorgas Case 2015-02 |
 |
|
The following patient was seen in the Medical ICU of Cayetano Heredia National Hospital in Lima, Perú.
 History: 41-year-old male transferred from another hospital with a 5-day history of fever to 38°C, headache, muscular pain in the lower extremities, nausea and vomiting. One day prior to admission his family noted jaundice, altered mental status, and reduced urine output. No history of rash, no respiratory symptoms, no genitourinary symptoms, no diarrhea. Since illness onset he had received IV fluids and antibiotics at the outside hospital; basic hematologic and biochemical lab testing (including liver function) on the 2nd day of illness had been within normal limits. History of childhood asthma.
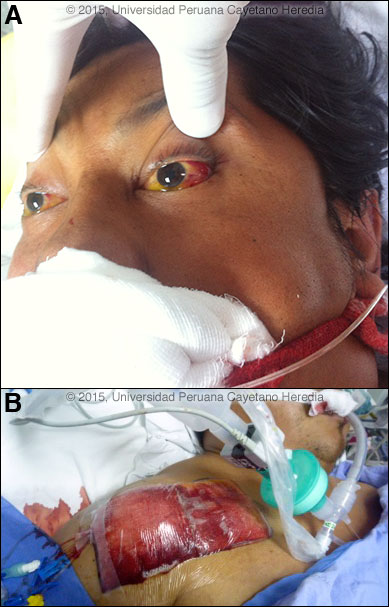
Epidemiology: Tailor, born in Cuzco in the highlands but lives in Lima. Returned home, 1 day prior to illness onset, from a car trip to Loreto in the Amazon jungle after an 11-day stay and had stayed for 2 previous days in San Martin, also in the jungle. Stayed on farms and used bed nets at night. Vaccinated against hepatitis B; took no malaria prophylaxis. Monogamous with wife, and no history of IV drug use or transfusions. No apparent exposure to rodents or animals. Physical Examination: BP 150/89, temperature 36.5°C, HR 122, respirations 34. Acutely ill and jaundiced with marked acidotic respiration and severe dehydration (capillary refill >2 seconds). Decreased level of consciousness with no meningeal or focal neurologic signs. Marked scleral icterus with spontaneous and extensive hemorrhage in conjunctivae [Image A], from all venipuncture sites, at the central catheter insertion site [Image B], mucosal areas including nose, oral cavity, from the nasogastric tube, and per rectum. Chest: clear. Cardiac exam: normal. Abdomen distended with normal bowel sounds and no peritoneal signs. Laboratory Examination (at admission on Day 5 of illness): Hb 16.5, WBC 9.0 (no differential), Platelets 188,000. PT 71.3 (N <13). Glucose 77. BUN 67 mg/dl. Creatinine 8.3 mg/dl. Albumin 3.3 (N>3.5). Bilirubin 8.0 (4.7 direct; 3.3 indirect). AST 13,300 (N <40). ALT 11,253 (N<40). LDH too high to measure. Alkaline phosphatase 176. CPK 825 (N<160). Urinalysis: protein 500mg/dl, RBC 95/hpf, WBC 0-5/hpf, glucose 250 mg/dl. Arterial blood gas: pH 7.3, HCO3 3.1. Serum lactate 7.3. Chest X-ray on admission was normal. Three thick smears negative for malaria; negative blood cultures. Negative Salmonella and Brucella agglutination tests.
|
|
Diagnosis: Yellow Fever. Fatal viral hemorrhagic fever.
 Discussion: IgM capture ELISA for yellow fever performed at the National Institute of Health of Perú was positive and dengue IgM was negative. PCR for dengue and yellow fever performed at the NAMRU-6 (U.S. Navy Medical Research Unit Lima) was negative on sample taken on Day 6 of illness, however, patients may only be viremic for 4-5 days. Serologies for HBV IgM core, HbsAg, HAV, HIV, and HTLV-1 were negative. Leptospirosis and rickettsia serologies are pending. Post-mortem liver biopsy histology is pending. Fulminant yellow fever is the true prototypical viral hemorrhagic fever (unlike Ebola Virus Disease in the current African outbreak) where patients bleed out and the liver dissolves [see recent review of yellow fever in J Clin Virol. 2014 Oct 24. pii:S1386-6532(14)00369-2]. Pathophysiological features are shared with several unrelated diseases except that the severity and extent of the hepatonecrosis is greatest in yellow fever patients. The hallmarks of the full yellow fever syndrome include the pathognomonic triad of hemorrhagic fever with jaundice and renal disease. Other viral hemorrhagic fevers may present with either jaundice or with renal disease, but the combination should always suggest yellow fever if there has been appropriate exposure according to the epidemiologic history.
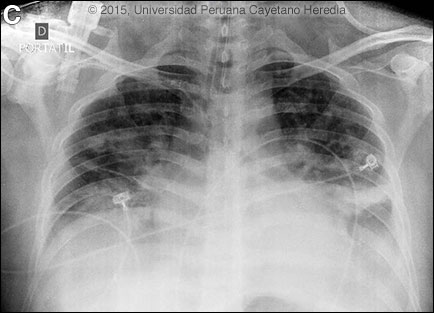
Mild cases of yellow fever cannot be distinguished clinically from falciparum malaria, leptospirosis, viral hepatitis, typhoid, rickettsial disease, hepatotoxin ingestion, or relapsing fever. More than 80% of yellow fever infections are symptomatic and the incubation period is usually 3 to 6 days. After an acute febrile illness with headache and myalgia without rash, which likely represents the peak viremia, there may be a period of remission (did not occur in this case). Fever may then resume with back pain, nausea, vomiting, and mental status changes progressing to the severe clinical syndrome already described above. Black vomitus (hematemesis) is commonly described. In fatal cases death usually occurs 6 to 10 days into the illness. Pathologically, yellow fever causes hepatocellular and Kupffer cell infection. There is mid-zonal hepatocellular necrosis with a minimal inflammatory response. So-called Councilman bodies and microvesicular fatty change is seen. A marked decrease of hepatic transaminases and bilirubin just before death likely represents near total destruction of functioning hepatocytes. In our patient, transaminases decreased from a peak of 300 times normal to 10 times normal (see below). In 2013 22 yellow fever cases, including 14 deaths (CFR: 63.6%), were confirmed in Perú, and in 2014 14 cases were confirmed, with 6 cases from San Martin and 3 cases from Loreto. Most Perúvian jungle departments report cases each year and in many recent years Perú has reported more cases of yellow fever than other countries in South America combined; this may reflect the excellent surveillance system. As with other flaviviruses there is no specific treatment for yellow fever, making prevention by use of 17D yellow fever vaccine (essentially 100% effective) imperative. While most individuals in endemic areas (Amazon basin and sub-Saharan Africa) have variable access to vaccines there is dramatic under-use of vaccine by travelers and expatriates. At least 10 unvaccinated short-term travelers to endemic areas have died of yellow fever since 1995. Data indicates that the number of unvaccinated travelers visiting risk areas is substantial. In Perú specifically, government vaccination programs cover the endemic areas, but residents of non-endemic areas who travel to the jungle are often poorly informed of the risk. The patient was started on ceftriaxone 2g/day, fluids, vasopressors, mechanical ventilation and dialysis. Chest X-ray on the 2nd hospital day showed an alveolar pattern consistent with early ARDS [Image C]. He died of multi-organ failure within 48 hours of admission and on Day 6 of illness. Prior to death Hb was 8.5, Creatinine 8.2, Albumin 2.3. AST/ALT decreased to 8,349/4,033, LDH to 28,873 mg/dl and Bilirubin to 5.7 mg/dl.
|