 |
Gorgas Case 2015-03 |
 |
|
The following patient was seen in the Outpatient Department of Cayetano Heredia National Hospital in Lima.

 History: 42-year-old male complaining of 2 years of skin problems that started with papular lesions on his left forearm, which in the following 6 months progressively involved the whole body. He complains of a burning sensation in the lesions, some nasal stuffiness and nosebleeds, as well as lower limb edema. He reports numbness in his forearms, hands, legs and feet. He has had several episodes of malaria and dengue and one episode of hepatitis of unknown etiology. He developed an erythematous rash on one occasion when he received antivenom for a snakebite. No history of chemical or irritant exposures. No trauma. No fever, weight loss or constitutional symptoms. No known contacts with a similar illness.
Epidemiology: Born in the highlands of Perú but has lived in the jungle for the last 10 years. Performs agricultural work. No history of sexually transmitted diseases or high-risk activity. No history of TB or TB contacts. Physical Examination: Afebrile. HR: 64/min; RR: 14/min. Good general condition. Lymph nodes of 1 to 1.5 cm in inguinal areas bilaterally. Skin: Diffuse, symmetric, erythematous cutaneous infiltrate over the face, trunk and limbs (sparing the axillae), midline of the back, scalp, palms and soles. Multiple non-tender erythematous papules and small superficial nodules on the forehead, cheeks, lips, chin, thorax, waist and limbs [Images A, B, C]. Neurologic Exam: Bilateral enlargement of common peroneal nerves and right posterior tibial nerve. Impairment of sensation on hands, forearms, legs, and feet, as well as over the skin lesions. Normal corneal reflex and no ocular inflammation. In addition, mild loss of motor function in the distribution of both ulnar nerves, both median nerves, and both common peroneal nerves. Rest of the examination was unremarkable. Laboratory Examination: Hematology and biochemistry unremarkable. A skin biopsy was obtained [see Discussion].
|
|
Diagnosis: Mycobacterium leprae. Multibacillary leprosy according to the WHO classification. Lepromatous leprosy (LL) leprosy according to the Ridley-Jopling classification.
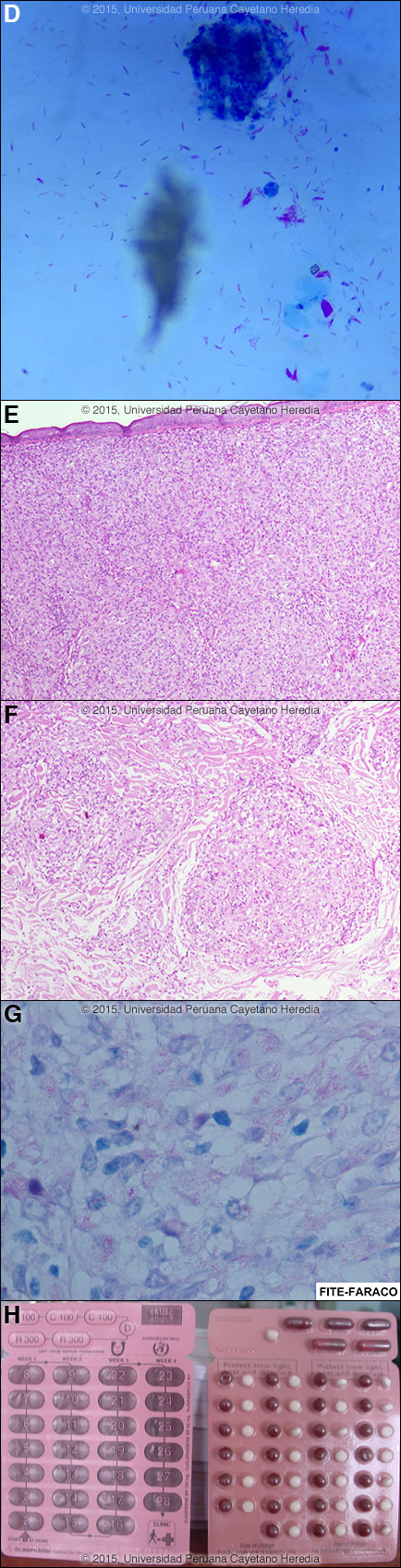
 Discussion: Slit skin smears were taken from both earlobes, both elbows (below the olecranon), and both knees (below the patella). Of the 6 smears, 3 had 6+ bacilli, and 3 had 5+ [Image D]. Slit skin smears are performed by making small (5 mm length, 2 mm depth) slits in pinched skin (to avoid bleeding), the edges of which are scraped. The material obtained is smeared on a clean slide and stained for AFB. The bacterial index ranges from zero (no bacilli in 100 oil-immersion fields) to 6+ (many clumps or globi or over 1000 bacilli in one field). Skin biopsy: Diffuse infiltrate of foamy histiocytes, separated from the epidermis by a grenz zone, that fills the whole dermis [Images E, F]. In a few areas where the infiltrate is more focal, the histiocytes are in elongated arrangements. The Fite-Faraco (tissue acid fast) stain shows massive amount of bacilli [Image G]. The findings are diagnostic of lepromatous leprosy. Leprosy is a disease of peripheral nerves and skin. Leprosy can be diagnosed clinically in any patient with simultaneous skin lesions and sensory loss over the lesions or in the distribution of the peripheral nerves. However, in early lepromatous cases, sensation may be normal over the lesions. Thus, with loss of sensation, a diagnosis of leprosy can be made; with intact sensation the diagnosis is possible but must be confirmed in some other way. This patient’s lesions are classic and typical. See Gorgas Cases 2013-09, 2012-02, and 2001-11 for typical skin findings in other patients. In lepromatous leprosy, bacilli invade the eyes, the nasal mucosa and intra nasal bones, the testes, and the small bones of the hand and feet. It is particularly important to examine the eyes carefully. For the purposes of determining treatment, the usual and most practical grading system is the WHO classification. For choosing the regimen, it matters only whether the patient has paucibacillary or multibacillary disease. Where no slit skins smears can be done, paucibacillary leprosy is defined as 5 or fewer skin lesions; multibacillary cases have 6 or more lesions. Paucibacillary disease usually presents with small numbers of hypopigmented macules or erythematous plaques with absent or reduced sensation and well-demarcated borders. Multibacillary disease is usually widespread at diagnosis with multiple macules, papules or nodules or infiltrated areas of skin with indistinct borders that may be non-anesthetic. The disease can be classified precisely in the immunologic sense using the traditional Ridley-Jopling classification. This describes a spectrum of disease ranging from tuberculoid leprosy (TT) with no AFB in lesions and good cell mediated immunity, to lepromatous leprosy (LL) with many AFB and poor cell-mediated immunity. This classification is especially useful in assessing risks and prognosis. The standard WHO regimen for paucibacillary disease is dapsone 100 mg/day unsupervised and rifampin 600 mg once per month directly observed for 6 months. For multibacillary disease, the long-standing recommendation is for patients to receive 100 mg dapsone and 50 mg clofazimine per day unsupervised and 600 mg rifampin and 300 mg of clofazimine directly observed once per month. The monthly dose of 300 mg of clofazimine in addition to the daily 50 mg dose is done just in case the patient is not compliant with the daily dose as the leprosy bacillus is very slowly multiplying. A standard WHO multibacillary dose-pack (provided free to endemic countries) is shown [Image H]; the instructions in English must be clarified for all healthcare staff and patients. WHO now recommends only 1 year of therapy for multibacillary cases [controversy discussed in Lancet. 2004 Apr 10;363(9416):1209-19], but some would treat those with high bacterial indices (4 to 6+) for the previously recommended 2 years due to higher relapse rates as we do here at our institution. For multibacillary disease in the USA and some other developed countries where the cost of rifampin is not limiting, the recommended first line regimen has for many years been 100 mg dapsone, 50 mg clofazimine, and 600 mg rifampin daily for 24 months. However, because the leprosy bacilli multiply very slowly (every 12-14 days), monthly rifampin is almost as effective as daily, there is no significant difference in the response rate. No comparative clinical or follow-up data on the different dosing regimens has been published and both are highly effective. However, many patients object to the severe cutaneous pigmentation that results from clofazimine therapy [see Gorgas Case 2005-04] and in the USA minocycline 100 mg/day in place of clofazimine is accepted as an alternative. However, evidence of the efficacy of minocycline’s anti-inflammatory activity against Type 2 reactions (see below) is not as substantial as the evidence for clofazimine. In adults, ofloxacin (400 mg/day) and clarithromycin (500 mg/day) are also sometimes used as a substitute for clofazimine in multi-dose regimens. The possibility of adverse effects of dapsone and clofazimine and of a lepra reaction should always be explained to a patient who is starting treatment and a reference text should be consulted prior to initiation of therapy by anyone not familiar with these. The most common reaction in multibacillary disease, occurring in about 50% of patients with lepromatous leprosy is a Type 2 reaction, which has as the most common presentation erythema nodosum leprosum (ENL). Type 2 reaction is due to an antigen-antibody-complement immune deposition in tissues, often accompanied by a severe systemic illness, and characterized by high levels of tissue and circulating TNF-alpha. ENL presents with fever together with many tender erythematous nodules [see Am J Trop Med Hyg. 2006;74(5):868-79]. Type 2 reaction may also produce to varying degrees, neuritis, uveitis, myositis, dactylitis, arthritis, periostitis, orchitis, lymphadenitis and nephritis accompanied by edema, arthralgia, and leukocytosis. ENL may occur in patients prior to therapy, during therapy and/or after therapy until the antigen load decreases markedly. ENL may present as repeated acute episodes or may be chronic and ongoing. ENL can be treated symptomatically if mild or with prednisone or thalidomide if severe [see Gorgas Case 2011-04]. In endemic areas, we usually examine the family contacts once a year for at least 5 years, and advise them to contact the health system in case they present any skin lesion or numbness. Acknowledgement: We thank Dr. Francisco Bravo, Gorgas Dermatology Professor, for interpretation and discussion of the skin biopsy.
|