 |
Gorgas Case 2019-04 |
 |
|
The following patient was seen in the outpatient clinic of the Infectious Diseases Department of the Hospital Cayetano Heredia during the Gorgas Diploma Course.
|
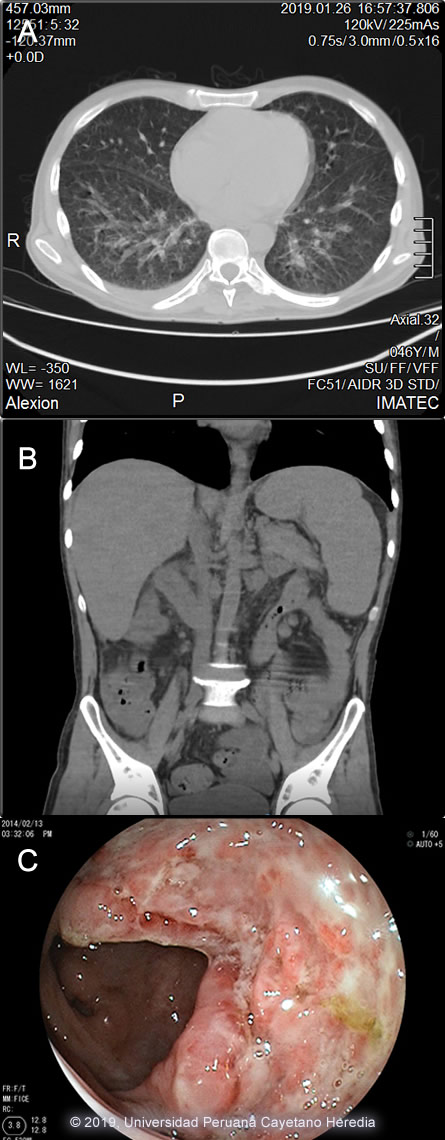 History: A 47-year-old male, HIV positive, presents with a 6-month history of abdominal pain, watery diarrhea, fever and weight loss. Epidemiology: Born in Chiclayo, resides in Jaen, Cajamarca but has been living in Lima for 8 months. Physical examination: T 36.7°C HR 120 RR 24 BP 90/50 Sat: 98 % (FiO2: 0.21) Laboratory result at admission: Hb: 7.7g/dL; WBC 1500 (0%bands, 87% neutrophils, 2% eosinophils, 8.5% lymphs); Platelets: 150 000; INR 2.35; Glucose 96 mg/dl; urea 33 mg/dl; creatinine 0.8 mg/dl; total bilirubin: 0.7 mg/dl; ALT 18 U/L (N=< 40); AST 78 U/L (N=<40); Alk phosphatase 406 mg/dl (N 15-60); GGT 400. LDH 1106. CD4 222; VL 361000. Negative tests for O & P including search for Strongyloides, Giardia and coccidian parasites, stool culture negative for bacteria. Negative Ziehl-Neelsen in a sputum sample as well as negative Gene Xpert MTB/RIF.
The chest x-ray showed bilateral interstitial infiltrates that were corroborated in the chest CT-scan [Image A]. An abdominal CT-scan showed visceromegaly [Image B]. A colonoscopy was performed, multiple colonic ulcers were observed scattered over the entire colon and ileum [Image C]. UPCH Case Editors: Carlos Seas, Course Director / Carlos McFarlane, Associate Coordinator UAB Case Editor: David O. Freedman, Course Director Emeritus / German Henostroza, Course Director |
|
Diagnosis: Progressive disseminated histoplasmosis (PDH) due to Histoplasma capsulatum
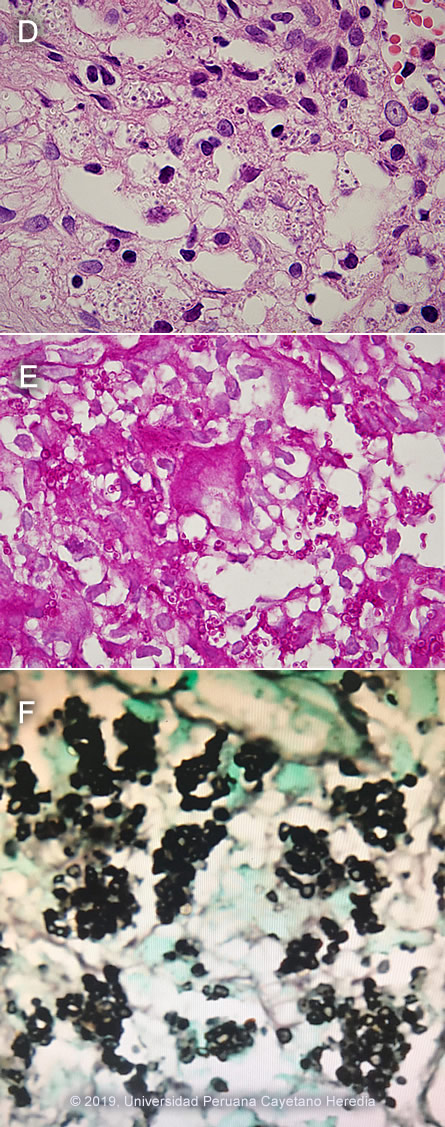
 Discussion: A biopsy of a colonic ulcer showed a dense histiocytic infiltrate within the lamina propia. [Image D]. Multiple oval-shape intracellular structures measuring 3-4 microns compatible with H. capsulatum were identified in a Periodic Acid Schiff stain [Image E] and in a Grocott stain [Image F]. The differential diagnosis includes paracoccidioidomycosis where the yeast cells would be of different sizes and show multiple peripheral budding mimicking a pilot´s wheel, and visceral leishmaniasis; not endemic in Peru, you would expect to see a kinetoplast. Definitive diagnosis in these cases is made by fungal (bone marrow, blood or tissue culture lymph nodes, skin or oral lesions) as a gold standard, these results are pending. Cultures are positive in about 75% of patients [AJTMH 2013;89:937]. Urinary Histoplasma antigen is very useful (97% sensitivity) but usually not available in developing countries [PLoS Negl Trop Dis 2018;12(10):e0006802]. Direct observation of yeast cells from blood samples has low sensitivity (10%), better results are obtained from bone marrow samples. H. capsulatum is a dimorphic endemic fungus of worldwide distribution but is most common in North America and Latin America. In highly endemic areas more than 80% of persons are infected by age 20, often sub-clinically. Bats, which often live in caves, are frequently infected. Acquisition is inhalational with principal sites of disease in lungs, lymph nodes, liver, spleen, bone marrow, adrenals and the GI tract. Histoplasmosis is endemic in Latin America including Peru, the fungus is predominantly found in jungle areas. The reported incidence among advanced HIV-infected patients varies from 8% in Panama to 42% in French Guyana, being the first or second opportunist infection in these patients [AIDS 2016;30:167]. A necropsy study of 16 patients who died with advanced AIDS in Lima, Peru found that 3 (19%) had disseminated histoplasmosis [Pathol Res Pract 2006;202:767]. Exogenous acquisition or reactivation of a latent foci, as in this case are the modes of acquisition. Histoplasmosis is the most common endemic mycosis among HIV-infected patients. Progressive disseminated histoplasmosis (PDH) as demonstrated by this patient is the most common manifestation of histoplasmosis in AIDS patients. PDH may be the first manifestation of advanced AIDS in endemic areas, a study among 89 patients with advanced AIDS in Colombia found 45 (51%) with PDH [AJTMH 2016;95:918]. Predictors of PDH were significant weight loss, hepato or splenomegaly, skin lesions and hematologic abnormalities (mainly pancytopenia), many of them seen in this patient. Pulmonary involvement is seen in about 50-70% with non-specific manifestations (dry cough, chest pain, dyspnea); 50-70% of patients have abnormal radiologic findings; diffuse interstitial infiltrates or reticulonodular infiltrates are the most common abnormal findings. PDH is associated with high mortality rates, a study among 101 confirmed cases in Guatemala found a crude mortality rate of 44% with a median survival time of 19 days [AJTMH 2017,97:42]; a Peruvian case series from a single center found that 59% (16) of 27 HIV-infected patients with histoplasmosis had PDH, with a mortality rate of 22% [Rev Chilena Infectol 2017;34:365-9]. Chronic diarrhea is a common clinical presentation among HIV-infected patients. A long list of potential causes includes bacteria, viruses, parasites, fungi and malignancies. A study conducted in Peru among 147 HIV-infected patients showed that Cryptosporidium sp. Giardia lamblia and bacterial agents were the most common etiologies of persistent-chronic diarrhea [J Infect Dis 2005;191(1):11, PMID 15592997]. Intestinal histoplasmosis has been reported among patients with advanced HIV-infection. Chronic diarrhea, wasting, vomiting, gastrointestinal bleeding, altered bowel habits and intestinal obstruction have been reported, multiple colonic ulcers are commonly observed [Int J Surg Pathol 2017;25(7):592]. Due to the non-specific nature of PDH presentation, patients with chronic diarrhea in endemic areas should be investigated for histoplasmosis. USA treatment guidelines [Clin Infect Dis 2007;45:807] recommend induction therapy with liposomal amphotericin B (or amphotericin B deoxycholate in persons with low risk for nephrotoxicity) for 1-2 weeks followed by itraconazole for at least 1 year, with dosing guided by measurement of serum drug levels. Suppressive therapy with itraconazole is stopped if patients have received one year of itraconazole therapy, have negative blood cultures, low levels of urinary antigen (<2ng/ml), a CD4 cell count >150 cells/mm3, and patients are on HAART. Initiation of HAART should not be delayed; IRIS is rare and usually not severe. Our patient had been started empirically on anti TB treatment with standard 4-drug therapy, which will not be continued, as there was no evidence of such an infection, and is currently receiving amphotericin deoxycholate. He will be started on ART after 2 weeks of induction therapy with amphotericin B. Failing a non-nucleoside reverse transcriptase reverse inhibitor-based regimen due to inconsistent adherence in developing countries compromises the selection of second-line regimens, a boosted protease inhibitor-based regimen will be selected in this patient [Lancet HIV 2017;4(10):e433]. |