Enhance Your Knowledge and Advance Your Research with the Latest CCTS Videos
New on the CCTS Website: NSF Grant Sample
Take Off on the CCTS Runway to Translational Research Success at July Forum
Community Focus: Birmingham Innovation Week
Mock NIH Study Section to Review K01 Grant Application
CCTS Launching New Round of R2T Kaizen Game
Calling All Bioinnovation Researchers and Entrepreneurs: Connect to Potential Customers, Funders via I-Corps@NCATS Method
Center for Genomic Medicine Deadline for Pilot Applications Fast Approaching
CCTS Provides Tips for Navigating a Changing Clinical Trial Landscape
CCTS Announces New Summer Dates for Popular i2b2 Training
Mark Your Calendar! I-Corps@NCATS Regional Short Course Informational Webinar
Building on Momentum: The Future of CCTS
CCTS Wants Your Ideas THIS WEEK
Master Mentoring with CCTS Spring Trainings
CCTS Translational Training Symposium Draws Rave Reviews
Join Us for 4th Annual CCTS Bioethics Forum
How to Work with the Media
CCTS and UAB Dept. of Radiology Relaunch Voucher Program
March Forum Features CTSA Career Development Expert Dr. Joel Tsevat
CCTS Alum, Experts in the News
Third Annual Translational and Transformative Informatics Symposium (ATTIS) Call for Abstracts
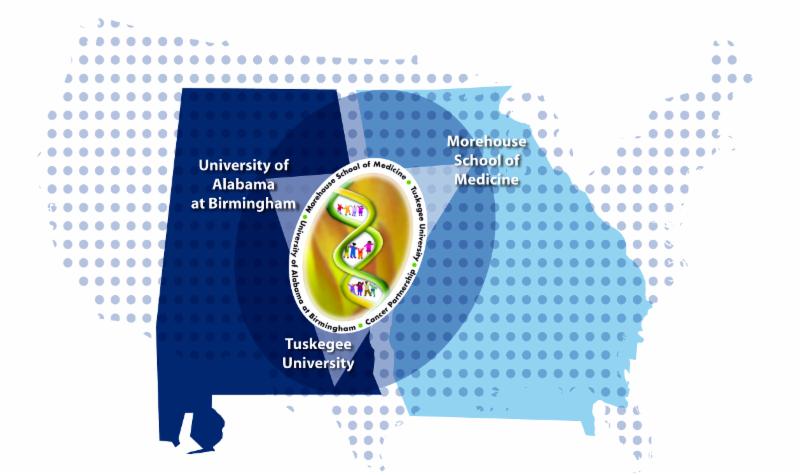
CCTS Partners UAB and Tuskegee Join Forces with Morehouse School of Medicine to Fund Pilot Projects Aimed at Reducing Cancer Disparities
Accessing Clinical Data for Research with i2b2 can help investigators:
- Determine study feasibility (sample cohort size)
- Identify potential participants for recruitment
- Perform simple data analyses of de-identified patient data
- Explore hypotheses for clinical studies/trials
In-person trainings have temporarily halted, but virtual trainings are available; you can still learn to use i2b2 to explore using UAB's electronic health record data for your research data needs! Keep up with all virtual learning opportunities through the CCTS Upcoming Events Page.
We strongly recommend attendees request i2b2 access prior to an i2b2 training session. Nonfaculty must obtain supervisor approval. Due to high demand, participation is limited to one session per attendee.
Jointly sponsored by the UAB Informatics Institute and the Center for Clinical and Translational Science (CCTS). To find out about the latest learning opportunities as soon as they are announced, subscribe to CCTS Digest and check our Events and Recurring Events web pages.
Welcome to the forum archive page where you can view previous CCTS Forum slides and video recordings. Other Important Resources:
Video Recording
CCTS Digest News Brief
Video Recording
CCTS Digest News Brief
Video Recording
CCTS Digest News Brief
Speaker: Scott J. Weir, PharmD, PhD, Director, Institute for Advancing Medical Innovation (IAMI) and Translational Research Professor, University of Kansas Medical Center
CCTS Digest News Brief
Speaker: Mark Yandell, PhD, Founder of IDbyDNA Inc. and Professor of Human Genetics at the University of Utah
March 1, 2017
Speaker: Erik Schwiebert, PhD, CEO and Chief Scientific Officer of DiscoveryBioMed, Inc.
Slide Deck
Video Recording
CCTS Digest News Brief
Speaker: Amy Hester, PhD, RN, BC (University of Arkansas for Medical Sciences)
Speakers: James J. Cimino, MD; Matthew Wyatt, MSHI; and Manfred Stapff, MD, PhD
Slide Deck
TriNetX Slide Deck
Video Recording
CCTS Digest News Brief
CCTS Digest News BriefSpeakers: Penny Jester, RN, MPH; David Redden, PhD; and Leslie Cooper
Slide Deck 1 (Redden)
Slide Deck 2 (Jester)
Slide Deck 3 (Jester)
Slide Deck 4 (Cooper)
Video Recording
Handouts:
JAMA Handout
NEJM Handout
GCP Training Policy Handout
CCTS News Handout
Moderator: David Redden, PhD
Panelists: Bruce Bunnell, PhD (Tulane University); Christopher Easley, PhD (Auburn University); Mark Gillespie, PhD (University of South Alabama); Emily Levitan, ScD (UAB); Hua Lu, MD, PhD (Tulane University); Vishnu Suppiramaniam, PhD, DVM (Auburn University); and Jianhua Zhang, PhD (UAB)
Video Recording
CCTS Digest News Brief
CCTS Digest News BriefSpeakers: David Chaplin, MD, PhD, Kenneth S. Saag, MD, MSc, Robin Lorenz, MD, PhD, Lisa Schwiebert, PhD, and Ann Smith, MPH
Dr. K. Saag Slide Deck
Dr. R. Lorenz Slide Deck
Ann Smith Slide Deck
Video Recording
Handouts:
Tools & Resources
Mentoring Contract
Individual Mentoring Development Plan (IMDP)
NRMN Powerpoint Slides
2016 UAB Mentoring Academy
Moderator: Jennifer Croker, PhD and David Redden, PhD
Panelists: Yabing Chen, PhD; Jianhua Zhang, PhD; Rita Cowell, PhD; and Mohammad Athar, PhD
Slide Deck
Video Recording
CCTS Digest News Brief
Speakers: Jake Chen, PhD and Elliot Lefkowitz, PhD
Chen Slide Deck
Lefkowitz Slide Deck
Video Recording
CCTS Digest News Brief
Speakers: UAB’s inaugural Informatics Institute Director Dr. James J. Cimino, UAB Health System Information Services IT Director and CCTS Informatics Co-Director Geoff Gordon, and CCTS Assistant Director of Informatics Matthew Wyatt.
Slide Deck
Video Recording
CCTS Digest News Brief
Speakers: David Redden, PhD and Stuart Frank, MD
Special Speakers: Madhuri Mulekar, PhD (University of South Alabama); Courtney Gamston, ScM, Pharm D (Auburn University); and Sonya Heath, MD (UAB)
Slide Deck
Video Recording
Speakers: Robert Kimberly, MD; Burt Nabors, MD; Elliot Lefkowitz, PhD; Michael Griswold, PhD (UMMC); Thomas Denney, PhD (Auburn); Mark Gillespie, PhD (USA); and Stuart Fran, MD
Slide Deck
Video Recording
Speakers: Jennifer Croker, PhD and David Redden, PhD December 3, 2015 (reprised December 16, 2015)
Slide Deck
Video Recording
Implementing Rigor and Transparency in NIH & AHRQ Research Grant and Career Development Applications
Notice Numbers: NOT-OD-16-011 and NOT-OD-16-012
On October 9, 2015, the NIH issued notices with updated grant submission and review instructions (please see links for full details). These changes are intended to enhance the reproducibility of research findings and are required with applications due January 25, 2016 and thereafter.
Find more information here >
“Our goals are to meet the needs of principal investigators while supporting the highest level of excellence in clinical and translational research, said CRU Nurse Manager Jolene Lewis, MSN, RN. “We are well trained in Good Clinical Practice Guidelines and offer highly skilled, safe, efficient, and compassionate care.” The CRU team’s experience encompasses the entire care spectrum, from healthy volunteers to intensive end-stage participants.
The CRU offers numerous services—both inpatient and outpatient—to support the needs of clinical trials and studies. These include:
• Medication administration/ Intravenous therapy
• Pharmacokinetic sampling
• Nursing assessment
• Patient education
• Treatment monitoring
• Point-of-care testing
• Cardiac & pulse oximetry monitoring
• Venipuncture/phlebotomy
• Questionnaire administration
The CRU clinic is the latest addition to our “First Wednesdays @ PCAMS” consultation clinics. CCTS also offers: (1) Biostatistics, Epidemiology, and Research Design (BERD); (2) Bionutrition; and (3) Biorepository. To learn more about our other clinics, see “CCTS Triple Feature Wednesdays.” To learn more about CRU or to request services, see our CRU page.